African Trypanosomiasis: Sleeping Sickness Overview
Understanding African sleeping sickness: transmission, symptoms, diagnosis, and treatment strategies.
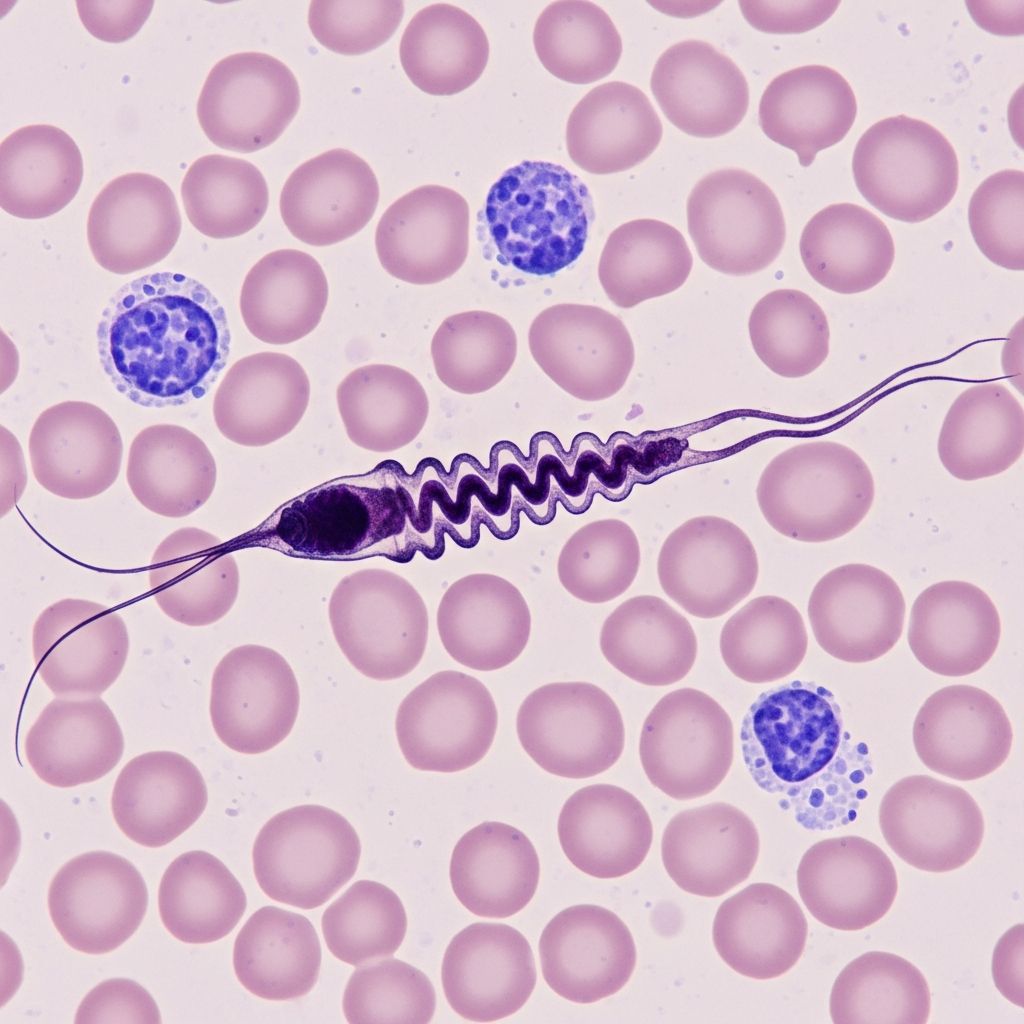
African Trypanosomiasis: Sleeping Sickness
African trypanosomiasis, commonly known as sleeping sickness, is a vector-borne parasitic disease endemic to sub-Saharan Africa that poses a significant public health threat. This disease is caused by protozoans of the genus Trypanosoma and is transmitted primarily through the bite of infected tsetse flies. The disease affects approximately 70 million people throughout sub-Saharan Africa and remains invariably fatal if left untreated or inadequately treated.
Cause and Transmission
African trypanosomiasis is caused by two subspecies of the parasitic protozoan Trypanosoma brucei: Trypanosoma brucei gambiense and Trypanosoma brucei rhodesiense. Humans are infected through the bite of an infected tsetse fly, a blood-feeding insect found exclusively in sub-Saharan Africa. The disease occurs in 36 sub-Saharan African countries, with transmission most common in rural areas where human-vector contact is frequent.
Trypanosoma brucei gambiense accounts for over 92% of reported cases and is found predominantly in West and Central Africa, causing a chronic form of the disease. Trypanosoma brucei rhodesiense, responsible for approximately 8% of reported cases, is found in 13 countries of eastern and southern Africa and causes an acute form of the disease with rapid progression.
Disease Progression and Stages
African trypanosomiasis progresses through two distinct clinical stages, each with characteristic symptoms and pathological features:
Hemolymphatic Stage (First Stage)
The first stage, known as the hemolymphatic or haemo-lymphatic phase, begins with an incubation period of 1–3 weeks for T. b. rhodesiense, though the incubation period is less precisely characterized in T. b. gambiense infections. During this stage, trypanosomes multiply in subcutaneous tissues, blood, and lymph, causing a range of non-specific, generalized symptoms.
Early manifestations typically include:
- Fever (intermittent)
- Severe headaches
- Joint pains and myalgia
- Itching and pruritus
- General weakness and malaise
- Fatigue and weight loss
- Lymphadenopathy (enlarged lymph nodes)
- Hepatosplenomegaly (enlarged liver and spleen)
These symptoms typically emerge one to three weeks after the tsetse fly bite. In fair-skinned individuals, a transient trypanosomal rash may develop, characterized by erythema, pruritus, and a targetoid appearance. A cutaneous chancre at the inoculation site may also be observed, though this is not always evident.
Meningoencephalitic Stage (Second Stage)
Without prompt treatment, the disease progresses to the second stage, known as the meningoencephalitic or neurological phase. During this stage, parasites cross the blood-brain barrier and invade the central nervous system, triggering a robust inflammatory response driven by interferon-λ (IFN-λ). This inflammatory cascade causes astrocyte activation, macrophage and T cell activation, and inflammatory infiltration of the brain vasculature.
Neurological symptoms characteristic of the second stage include:
- Behavioral changes and personality alterations
- Confusion and disorientation
- Sensory disturbances and numbness
- Poor coordination (ataxia)
- Sleep cycle disturbances and insomnia
- Progressive mental deterioration
Sleep disturbance is particularly prominent and gives the disease its common name, “sleeping sickness,” though the sleep cycle abnormality is complex and not simply characterized by excessive daytime somnolence. Without treatment, progressive mental deterioration leads to coma, systemic organ failure, and death.
Disease Variants and Clinical Differences
The two subspecies of Trypanosoma brucei present distinctly different clinical courses and epidemiological patterns:
| Characteristic | T. b. gambiense (West African) | T. b. rhodesiense (East African) |
|---|---|---|
| Percentage of Cases | Over 92% | Approximately 8% |
| Geographic Distribution | West and Central Africa | Eastern and Southern Africa (13 countries) |
| Disease Course | Chronic, protracted | Acute, rapidly progressive |
| Symptom Onset | Weeks to months to years | Few weeks to months |
| CNS Invasion | Slower progression | Rapid, multi-organ invasion |
| Untreated Mortality Timeline | Several years (average 3 years) | Weeks to months |
T. b. rhodesiense is the acute form of the disease, with symptoms emerging within weeks and death occurring within months if untreated. This subspecies is significantly more virulent and faster-developing than T. b. gambiense. In contrast, individuals infected with T. b. gambiense may remain asymptomatic or have mild symptoms for months or even years before advancing to the meningoencephalitic stage, where successful treatment becomes increasingly difficult.
Parasite Biology and Immune Evasion
Trypanosomes employ sophisticated mechanisms to evade the human immune system. The parasite is surrounded by a coat composed of variant surface glycoproteins (VSG), which protect the parasite from lytic factors present in human plasma. The host’s immune system recognizes these glycoproteins and produces different antibodies (IgM and IgG) in response. However, the parasites can alter their surface glycoproteins, allowing them to evade antibody recognition and persist in the host.
During the course of infection, slender trypanosomes can transform into a stumpy form when they reach adequate parasitemia levels. This transformation is triggered by increased secretion of stumpy induction factor (SIF) by the parasites. While stumpy forms are short-lived, they are more capable of infecting the tsetse fly vector than slender parasites, facilitating the parasite’s transmission cycle.
Diagnosis
Accurate diagnosis of African trypanosomiasis is essential for initiating appropriate treatment. The disease is diagnosed through detection of the parasite in biological samples, with different diagnostic approaches used depending on the stage of disease:
- Blood smear examination: Direct microscopic examination of blood for motile trypanosomes is a common diagnostic method, particularly useful in early-stage disease when parasitemia levels are high.
- Lymph node fluid examination: Aspiration and examination of lymph node fluid can reveal trypanosomes, especially in patients with lymphadenopathy.
- Lumbar puncture (cerebrospinal fluid analysis): This procedure is often necessary to detect parasites in the cerebrospinal fluid and to differentiate between first-stage and second-stage disease. CSF examination is particularly important for confirming CNS involvement and guiding treatment decisions.
Lumbar puncture is a critical diagnostic tool because clinical features alone may not reliably distinguish between hemolymphatic and meningoencephalitic stages, particularly in cases where neurological symptoms may coexist with parasitemia in the blood and lymphatic systems.
Treatment Considerations
The treatment of African trypanosomiasis depends on the disease stage, subspecies involved, and clinical presentation. Various antimicrobial agents are available, each with specific indications and limitations. Early diagnosis and prompt initiation of appropriate therapy are crucial for preventing progression to the neurological stage and improving clinical outcomes. Treatment failure can occur, particularly if CNS involvement is advanced or if treatment is delayed.
Prognosis and Outcomes
Without treatment, African trypanosomiasis is invariably fatal. An untreated infection with T. b. rhodesiense will cause death within months, while an untreated infection with T. b. gambiense will cause death after several years. Damage caused in the neurological phase is irreversible, underscoring the importance of early detection and treatment.
The clinical endpoint of either form of HAT, if untreated, is coma followed by death. Rare cases of self-cure have been reported, but these are exceptional and cannot be relied upon. For individuals infected with T. b. gambiense, the chronic nature of infection means that individuals may unknowingly harbor the parasite and progress to advanced disease before seeking treatment, at which point successful therapeutic intervention becomes significantly more challenging.
Disease Burden and Public Health Impact
African trypanosomiasis is classified as a neglected tropical disease and represents a significant public health threat, particularly in rural areas of sub-Saharan Africa. With an estimated 70 million people at risk throughout the region, the disease’s burden extends beyond direct mortality to include substantial economic and social costs. The disease disproportionately affects impoverished populations in endemic areas, where access to diagnosis and treatment may be limited.
Special Considerations in Travelers
While African trypanosomiasis is endemic to sub-Saharan Africa, the disease can be observed outside of endemic areas in travelers who have been exposed to infected tsetse flies. HAT in travelers may present differently from cases in endemic populations, often manifesting with more pronounced cutaneous chancres and internal organ involvement. Clinicians should maintain awareness of this possibility when evaluating febrile illness with headache, joint pain, and lymphadenopathy in returning travelers from sub-Saharan Africa.
Frequently Asked Questions
Q: How is African trypanosomiasis transmitted?
A: African trypanosomiasis is transmitted through the bite of an infected tsetse fly. The disease is not transmitted from person to person through casual contact, though rare congenital transmission and transmission through blood transfusion have been documented.
Q: What is the difference between West African and East African sleeping sickness?
A: West African sleeping sickness, caused by T. b. gambiense, is a chronic disease that may progress slowly over years without symptoms. East African sleeping sickness, caused by T. b. rhodesiense, is an acute disease with rapid onset of symptoms and progression to neurological involvement within weeks to months. East African disease is more virulent and fatal more quickly if untreated.
Q: Can African trypanosomiasis be cured?
A: Yes, African trypanosomiasis can be cured with appropriate antimicrobial treatment, particularly when diagnosed and treated early in the hemolymphatic stage. However, treatment is more challenging once the disease has progressed to the meningoencephalitic stage, and damage caused in the neurological phase may be irreversible.
Q: How long can someone live with untreated African trypanosomiasis?
A: Without treatment, T. b. rhodesiense infection typically causes death within weeks to months, while T. b. gambiense infection may result in death after several years. The disease is invariably fatal without appropriate treatment.
Q: What should travelers do to prevent African trypanosomiasis?
A: Travelers to endemic areas should minimize exposure to tsetse flies by wearing protective clothing, using insect repellent, avoiding certain areas during peak fly activity, and seeking prompt medical evaluation if symptoms develop after potential exposure. No vaccine is currently available for prevention.
References
- African Trypanosomiasis (Sleeping Sickness) — Center for Emerging and Re-emerging Infectious Diseases, University of Washington. Accessed 2026. https://cerid.uw.edu/research/diseases-we-study/african-trypanosomiasis
- Trypanosomiasis, Human African (Sleeping Sickness) — World Health Organization. 2024. https://www.who.int/news-room/fact-sheets/detail/trypanosomiasis-human-african-(sleeping-sickness)
- Human African Trypanosomiasis (Sleeping Sickness) — National Center for Biotechnology Information (NCBI), National Institutes of Health. 2024. https://www.ncbi.nlm.nih.gov/books/NBK535413/
- Human African Trypanosomiasis (Sleeping Sickness) — Frontiers in Tropical Diseases. 2023. https://www.frontiersin.org/journals/tropical-diseases/articles/10.3389/fitd.2023.1087003/full
- Sleeping Sickness: Symptoms, Treatment, Prevention — Institut Pasteur. 2024. https://www.pasteur.fr/en/medical-center/disease-sheets/sleeping-sickness
Read full bio of Sneha Tete