Allergy To Ecamsule: What You Need To Know
Understanding ecamsule allergies: symptoms, causes, diagnosis, and safe sun protection strategies for sensitive skin.
Allergy to Ecamsule
Ecamsule, also known as Mexoryl SX or terephthalylidene dicamphor sulfonic acid, is a chemical ultraviolet (UV) filter used in sunscreens to protect against UVA radiation. While effective, it can trigger allergic reactions, particularly photocontact dermatitis in sensitised individuals. This article explores the nature of ecamsule, its role in sun protection, associated allergies, diagnostic methods, treatment options, and prevention strategies.
What is ecamsule?
Ecamsule is a modern organic sunscreen agent specifically designed to absorb UVA rays, which penetrate deeper into the skin and contribute to photoageing, wrinkles, and skin cancer risk. Introduced relatively recently to the sunscreen market, ecamsule offers superior photostability compared to older UVA filters like avobenzone. Photostability refers to the ingredient’s ability to maintain efficacy under prolonged UV exposure without breaking down.
Unlike avobenzone, which can degrade by up to 50% after hours of sun exposure, ecamsule retains about 60% of its protective properties even after extended UV irradiation. To enhance its performance, ecamsule is often combined with stabilisers such as octocrylene and avobenzone in formulations like Anthelios SX. In vitro studies demonstrate these combinations can provide effective protection for up to 5 hours.
Chemically, ecamsule is terephthalylidene dicamphor sulfonic acid, a water-soluble compound that forms a stable film on the skin surface. It is primarily available in L’Oréal products in the US and select international markets. Its approval by the FDA was based on safety studies showing it is generally well-tolerated, with infrequent mild side effects.
Who gets allergy to ecamsule?
Allergic reactions to ecamsule are uncommon but can affect anyone using products containing it, particularly those with sensitive skin or a history of contact dermatitis. It is more prevalent in individuals frequently applying sunscreens, such as outdoor workers, athletes, or those in sunny climates. Sensitisation often occurs after repeated exposure, even if initial use was uneventful.
People with pre-existing conditions like eczema, lupus erythematosus, or polymorphous light eruption (PMLE) may be at higher risk, as ecamsule-containing sunscreens have been studied for photoprotection in these groups. While effective for lupus patients, isolated cases of allergy have been reported. Children, fair-skinned individuals, and those using high-SPF broad-spectrum sunscreens are also susceptible.
Clinical features of allergy to ecamsule
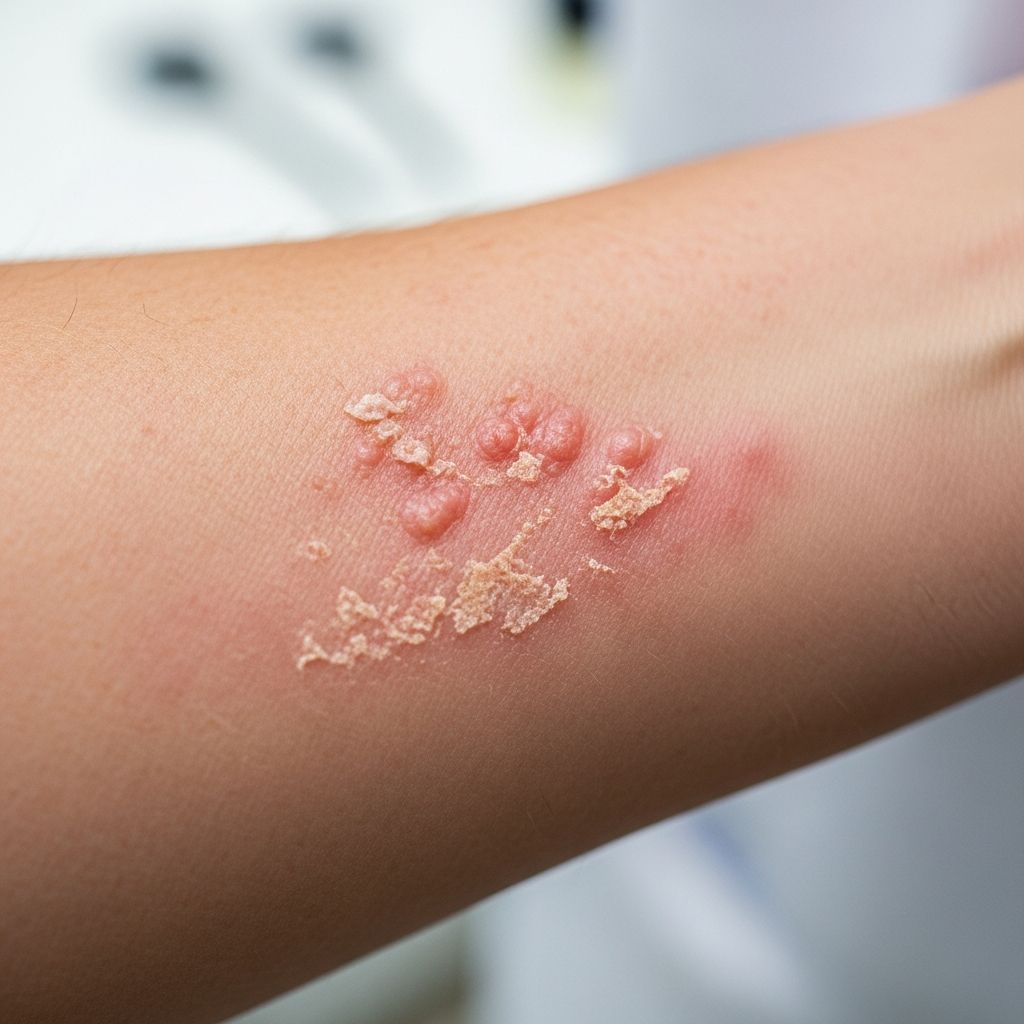
The hallmark of ecamsule allergy is photocontact dermatitis, a type IV hypersensitivity reaction triggered by UV-exposed ecamsule on the skin. Symptoms appear only on sun-exposed areas, typically 24-48 hours after application and UV exposure. Common manifestations include:
- Redness (erythema) and inflammation
- Itching (pruritus) and burning sensation
- Dry, scaly skin or acne-like eruptions
- Papules, vesicles, or blisters in severe cases
- Oedema (swelling) on face, neck, hands, or V-neck area
Unlike irritant contact dermatitis, which occurs without UV light, photocontact reactions are photoallergic, meaning UV activation is required. Mild reactions resemble sunburn, while severe ones mimic acute eczema. Systemic symptoms are rare but can include hives or, exceptionally, anaphylaxis with breathing difficulty[10].
Reactions are usually confined to application sites but can spread with continued exposure. Chronic use without awareness may lead to persistent dermatitis.
Potential complications
Untreated ecamsule photocontact dermatitis can lead to secondary bacterial infections from Staphylococcus aureus, post-inflammatory hyperpigmentation, or scarring. Prolonged inflammation may exacerbate underlying conditions like rosacea or exacerbate photoageing. In rare cases, repeated sensitisation increases reactivity to other UV filters, complicating sun protection.
How is the diagnosis of allergy to ecamsule made?
Diagnosis begins with a detailed history: timing of symptoms post-sunscreen application and sun exposure, product ingredients, and prior reactions. Physical exam reveals characteristic photo-distribution patterns.
Patch testing is the gold standard. Small amounts of diluted ecamsule (typically 0.1-1% in petrolatum or aqueous solution) are applied to the upper back under occlusion for 48 hours. Sites are read at 48 and 96 hours. A positive reaction shows erythema, papules, or vesicles (graded + to +++). For photocontact allergy, photopatch testing is essential: duplicate patches are applied, one irradiated with UVA (5 J/cm²), both read at 48/96 hours. Reaction only on the irradiated patch confirms photoallergy.
Self-testing is discouraged without medical advice but involves applying a coin-sized amount to the inner arm for several days, monitoring daily. Phototesting may supplement if patch tests are negative. Differential diagnosis includes irritant dermatitis, PMLE, solar urticaria, or airborne contact dermatitis.
What is the treatment of allergy to ecamsule?
Immediate management: discontinue the product, rinse skin with cool water, avoid sun exposure until resolution (several days to weeks). Cool compresses soothe acute inflammation.
Topical treatments:
- Emollients and fragrance-free moisturisers to restore barrier
- Low-potency topical corticosteroids (e.g., hydrocortisone 1%) for mild cases
- Medium-potency steroids (e.g., triamcinolone) for moderate-severe, tapered over 1-2 weeks
- Calcineurin inhibitors (tacrolimus) for facial/periorbital areas
Systemic treatments: Oral antihistamines (loratadine, cetirizine) for itching; short-course oral corticosteroids (prednisone 0.5-1 mg/kg) for widespread reactions. Antibiotics if infected.
For refractory cases, UV therapy or omalizumab may be considered, though primarily for solar urticaria.
What is the likely outcome of allergy to ecamsule?
With prompt avoidance and treatment, most reactions resolve fully within 1-4 weeks without sequelae. Sensitisation may persist lifelong, necessitating permanent avoidance of ecamsule-containing products. Cross-reactivity with other benzylidene camphor derivatives is possible but rare. Long-term prognosis is excellent with proper sun protection alternatives.
How can allergy to ecamsule be prevented?
Prevention centres on avoidance:
- Read labels: Avoid ‘ecamsule’, ‘Mexoryl SX’, ‘terephthalylidene dicamphor sulfonic acid’
- Choose mineral sunscreens with zinc oxide/titanium dioxide, less allergenic
- Opt for hypoallergenic, fragrance-free, broad-spectrum SPF 30+ products
- Perform pre-use testing under medical guidance
- Wear protective clothing, hats, seek shade during peak UV hours
Table of alternative UV filters:
| Filter Type | Examples | Allergenicity Risk |
|---|---|---|
| Physical/Mineral | Zinc oxide, Titanium dioxide | Low |
| Chemical UVA | Tinosorb S, Tinosorb M | Low-moderate |
| Chemical UVB | Octinoxate (limited) | Moderate |
Consult dermatologists for personalised recommendations.
Frequently Asked Questions
What are the first signs of ecamsule allergy?
Redness, itching, and burning on sun-exposed skin 24-48 hours after sunscreen application.
Can ecamsule allergy develop suddenly?
Yes, after repeated exposure even if previously tolerated.
Is patch testing safe for diagnosing?
Yes, when performed by specialists; photopatch testing confirms photoallergy.
Are mineral sunscreens always safe?
Generally yes, but rare allergies occur; test new products.
How long do symptoms last?
1-4 weeks with treatment and avoidance.
References
- Allergy to ecamsule — DermNet NZ. 2023. https://dermnetnz.org/topics/allergy-to-ecamsule
- Ecamsule: Uses, Interactions, Mechanism of Action — DrugBank Online. 2024-01-15. https://go.drugbank.com/drugs/DB09534
- A Comprehensive Guide to Contact Dermatitis from Sunscreens — Austin Allergist. 2024. https://www.austinallergist.com/a-comprehensive-guide-to-contact-dermatitis-from-sunscreens-causes-symptoms-and-solutions/
- Potential allergenicity of commonly sold high SPF broad spectrum sunscreens — PMC (NCBI). 2019-10-22. https://pmc.ncbi.nlm.nih.gov/articles/PMC6831754/
- Sunscreen allergy — DermNet NZ. 2023. https://dermnetnz.org/topics/sunscreen-allergy
- Photosensitivity Reactions — Merck Manuals. 2024. https://www.merckmanuals.com/home/skin-disorders/sunlight-and-skin-damage/photosensitivity-reactions
Read full bio of Sneha Tete