CAR T-Cell Therapy: Advanced Cancer Treatment
Understanding CAR T-cell therapy: A revolutionary immunotherapy for blood cancers and beyond.
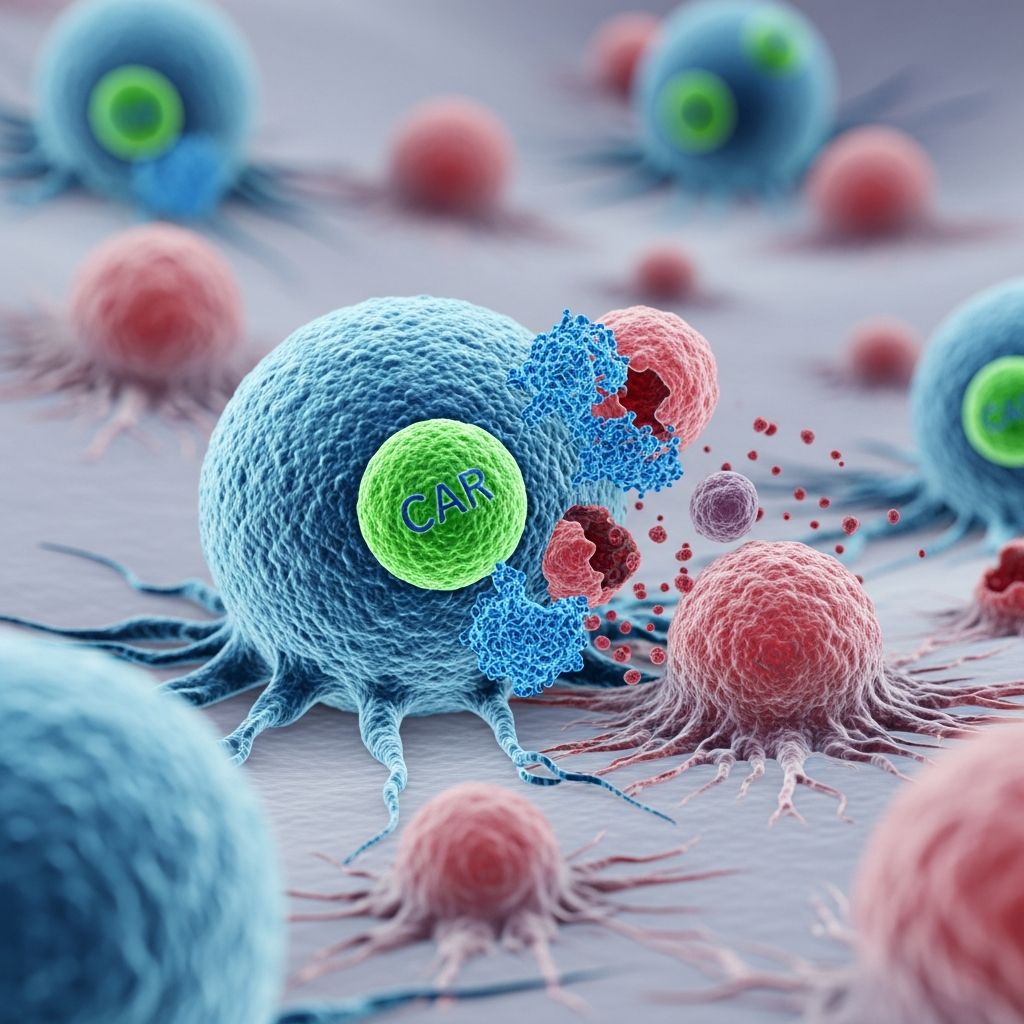
What is CAR T-Cell Therapy?
CAR T-cell therapy stands for chimeric antigen receptor T-cell therapy, representing one of the most innovative approaches to cancer treatment in recent decades. This groundbreaking immunotherapy works by harnessing the power of a patient’s own immune system to fight cancer cells. Unlike traditional chemotherapy or radiation, which attack all rapidly dividing cells indiscriminately, CAR T-cell therapy is specifically engineered to target cancer cells while minimizing damage to healthy tissue.
The therapy begins with a straightforward but sophisticated process. T-cells, which are specialized white blood cells that normally help your body fight infections and diseases, are extracted from a patient’s blood through a procedure called apheresis. These cells are then sent to a laboratory where they undergo genetic modification. Scientists insert a special gene into the T-cells that codes for a chimeric antigen receptor, enabling these cells to recognize and bind to specific markers found on cancer cells. Once this genetic modification is complete, the engineered T-cells are expanded in large numbers in the laboratory before being reinfused back into the patient’s body.
How CAR T-Cell Therapy Works
The mechanism of action behind CAR T-cell therapy involves several carefully orchestrated steps that transform ordinary immune cells into precision cancer fighters. When CAR T-cells are introduced back into the patient’s body, they circulate through the bloodstream searching for cells displaying the target antigen that matches their newly acquired receptor. Once these engineered cells locate and bind to their target cancer cells, a cascade of immune responses is triggered.
Upon binding to target cells, CAR T-cells proliferate rapidly and produce powerful cytotoxic molecules including interferon-gamma, granulocyte macrophage colony-stimulating factor, interleukin-10, and interleukin-6. These cytokines work in concert to destroy the recognized cancer cells through multiple mechanisms. The therapy essentially educates the immune system to become a highly specialized cancer-fighting force, capable of distinguishing between malignant cells and healthy tissue with remarkable precision.
FDA-Approved Uses
Since initial FDA approval in 2017, CAR T-cell therapy has been approved for treating several types of blood cancers, particularly hematological malignancies where this approach has demonstrated exceptional efficacy.
Current FDA-approved applications include:
- Diffuse large B-cell lymphoma (DLBCL)
- Follicular lymphoma
- Mantle cell lymphoma
- Acute lymphoblastic leukemia in younger patients
- Multiple myeloma
These approvals represent a major advancement in cancer treatment, offering hope to patients who may have limited options with conventional therapies. The expansion of approved indications demonstrates the therapy’s versatility in treating different blood cancers, each with distinct biological characteristics.
The CAR T-Cell Therapy Process
Pre-Treatment Evaluation
Before beginning CAR T-cell therapy, patients undergo comprehensive evaluation to ensure they are appropriate candidates. Healthcare providers assess overall health status, disease burden, and functional capacity. Importantly, unlike stem cell transplantation where patients typically must be in remission, CAR T-cell therapy can be administered to patients with active disease, making it a viable option for individuals with aggressive or resistant cancers.
Cell Collection and Engineering
The first step involves collecting T-cells from the patient’s blood through apheresis, a procedure similar to donating blood plasma. During apheresis, blood is drawn from the patient, the T-cells are separated from other blood components, and the remaining blood is returned to the patient. This typically takes several hours in an outpatient setting.
Once collected, the T-cells are transported to a specialized laboratory where genetic modification occurs. Scientists use sophisticated molecular techniques to insert the CAR gene into the T-cells, permanently altering their genetic makeup to enable cancer recognition and destruction. The engineered cells are then cultured and expanded, sometimes taking several weeks to generate sufficient quantities for therapeutic use.
Lymphodepleting Chemotherapy
Before CAR T-cells are reinfused, patients receive a brief course of lymphodepleting chemotherapy, typically less intensive than standard cancer treatments. This chemotherapy serves a critical purpose: it suppresses the patient’s existing immune system, creating biological space for the newly engineered CAR T-cells to expand and proliferate effectively. This preparatory step significantly enhances the therapeutic efficacy of the CAR T-cells, allowing them to thrive and multiply in numbers sufficient to combat the cancer.
Recent advances have made lymphodepleting chemotherapy more flexible, with some patients now able to receive this preparation in an outpatient setting, reducing hospitalization time and improving quality of life during treatment.
CAR T-Cell Infusion and Hospitalization
Once the engineered T-cells have been sufficiently expanded, they are reinfused into the patient’s bloodstream. This infusion typically occurs during a hospital admission, with most patients expected to remain hospitalized for 7-10 days, though this timeframe is decreasing as medical teams gain experience with the therapy. The infusion itself is relatively straightforward, delivered through an intravenous line, but close monitoring is essential during the initial period following reinfusion.
Benefits of CAR T-Cell Therapy
CAR T-cell therapy offers several distinct advantages over traditional cancer treatments. The therapy provides targeted destruction of cancer cells while preserving healthy tissue, resulting in fewer side effects compared to conventional chemotherapy or radiation. For many patients, particularly those with resistant or relapsed cancers, CAR T-cell therapy offers the possibility of durable remission or potentially curative responses.
The ability to treat patients with active disease, rather than requiring remission first, expands the potential patient population and offers hope to those with aggressive malignancies. Additionally, the immunological memory generated by CAR T-cells means the engineered cells can persist for years, providing long-term protection against cancer recurrence.
Potential Side Effects and Complications
Cytokine Release Syndrome
The most significant side effect associated with CAR T-cell therapy is cytokine release syndrome (CRS), which occurs when the engineered T-cells activate and release large amounts of inflammatory cytokines. This immune activation, while therapeutically beneficial against cancer, can produce flu-like symptoms ranging from mild to severe, including fever, chills, fatigue, and body aches.
In more severe cases, CRS can cause increased vascular permeability, allowing fluid to leak from blood vessels into surrounding tissues, resulting in decreased cardiac output and intravascular volume depletion. Most cases of CRS are manageable with supportive care and anti-inflammatory medications, though severe cases require intensive monitoring in a hospital setting.
Neurological Effects
Some patients experience CAR T-cell related encephalopathy syndrome (CRES), characterized by confusion, difficulty speaking or writing, hallucinations, or even seizures. While these neurological complications are generally reversible, they require prompt recognition and treatment.
Other Potential Side Effects
Additional side effects may include cytopenias (low blood cell counts), infections due to immune suppression, and infusion-related reactions. Low blood cell counts can persist for some time following treatment, requiring careful monitoring and sometimes necessitating blood transfusions or supportive care measures.
Beyond Cancer: Emerging Applications
While CAR T-cell therapy is currently approved for blood cancers, research is expanding into non-malignant conditions with promising results. Phase 2 and Phase 3 clinical trials are underway examining CAR T-cell therapy for systemic lupus erythematosus (SLE), an autoimmune disease where B-cells play a pathogenic role.
Early trial results show remarkable promise. In SLE patients treated with anti-CD19 CAR T-cells targeting B-cells, nephritis (kidney inflammation) ceased in all treated patients, with arthritis, fatigue, and pulmonary symptoms resolving. Notably, patients were able to discontinue all disease-modifying antirheumatic drugs and corticosteroids, achieving drug-free remission. Similar therapeutic approaches are being investigated for other autoimmune conditions including multiple sclerosis, potentially revolutionizing treatment for these previously difficult-to-manage diseases.
Who May Benefit from CAR T-Cell Therapy
Ideal candidates for CAR T-cell therapy include patients with FDA-approved blood cancers who have either failed conventional treatments or present with aggressive disease requiring immediate intervention. Patients should have adequate organ function to tolerate the lymphodepleting chemotherapy and potential side effects. Importantly, recent experience has expanded the eligible population to include older patients and those with comorbidities when the cancer itself poses greater risk than the therapy.
Patients must be able to commit to close follow-up care and monitoring during and after treatment, as complications can emerge during the hospitalization period and the months following CAR T-cell infusion.
Treatment Trends and Future Directions
Expanding Access and Outpatient Administration
A major focus for advancing CAR T-cell therapy involves increasing accessibility and moving treatment toward the outpatient setting. Currently, specialized academic medical centers primarily administer CAR T-cell therapy, creating barriers for many patients who lack convenient access to these institutions. Researchers and clinicians are developing strategies to streamline the production process, reducing the time required to manufacture CAR T-cells and potentially enabling more hospitals to offer this treatment.
As more patients can receive lymphodepleting chemotherapy in outpatient settings and as manufacturing becomes more efficient, the vision is for patients to eventually receive CAR T-cell therapy without requiring prolonged hospitalization, making the therapy more practical and affordable for broader patient populations.
Clinical Trial Expansion
The surge in CAR T-cell approval has resulted in numerous clinical trials investigating novel applications. Cleveland Clinic Cancer Institute alone expects to treat 150 patients with CAR T-cell therapy in 2024, with demand projected to more than double by 2028. This explosive growth reflects both the clinical success of the therapy and the growing recognition of its transformative potential.
Frequently Asked Questions
Q: How long does the CAR T-cell manufacturing process take?
A: The manufacturing process typically takes several weeks to generate sufficient quantities of engineered cells for therapeutic use. However, ongoing research aims to accelerate this timeline, potentially reducing manufacturing duration and increasing access to treatment.
Q: Can CAR T-cell therapy cure cancer?
A: While CAR T-cell therapy offers the possibility of durable remission and potentially curative responses, particularly in blood cancers, individual outcomes vary. Some patients achieve long-term remission, while others may experience recurrence requiring additional treatment.
Q: How long do patients remain hospitalized?
A: Most patients are hospitalized for 7-10 days following CAR T-cell infusion, though this timeframe is decreasing as medical teams gain experience and as more aspects of treatment move to the outpatient setting.
Q: What makes CAR T-cell therapy different from stem cell transplantation?
A: Unlike stem cell transplantation where patients must typically be in remission and have excellent functional status, CAR T-cell therapy can be administered to patients with active disease and more variable health status, expanding the eligible patient population.
Q: Are there any contraindications to CAR T-cell therapy?
A: Contraindications include severe organ dysfunction, uncontrolled infections, or inability to tolerate lymphodepleting chemotherapy. Each patient requires individualized evaluation to determine suitability for treatment.
References
- CAR T-cell Therapy Management — Cleveland Clinic. 2024. https://my.clevelandclinic.org/podcasts/cancer-advances/car-t-cell-therapy-management
- Cellular and Immunotherapy Service Provides Patients with Standard of Care and Clinical Trial Opportunities for Novel Therapies — Cleveland Clinic. 2024. https://consultqd.clevelandclinic.org/new-cellular-and-immunotherapy-service-provides-patients-with-standard-of-care-and-clinical-trial-opportunities-for-novel-therapies
- The Evolution of CAR T Therapy and Its Use in Systemic Lupus Erythematosus — Cleveland Clinic. 2024. https://consultqd.clevelandclinic.org/the-evolution-of-car-t-therapy-and-its-use-in-systemic-lupus-erythematosus
- Insights About CAR T-Cell Therapy — Cleveland Clinic. 2024. https://consultqd.clevelandclinic.org/insights-about-car-t-cell-therapy
- The Future of CAR T-Cell Therapy — Cleveland Clinic Lerner Research Institute. 2024. https://www.lerner.ccf.org/news/article/?title=The+Future+of+CAR+T-Cell+Therapy&id=4bbac50925eba0486400c63e69cb9d4098333587
- Advancing CAR T-cell therapies: Preclinical insights and clinical applications — PubMed Central. 2024. https://pubmed.ncbi.nlm.nih.gov/39289094/
- The Complex Equation of Outpatient CAR T-Cell Therapy — Journal of Oncology Practice. 2025. https://ascopubs.org/doi/10.1200/OP-25-00291
Read full bio of Sneha Tete