Dermoid Cyst Pathology: Clinical, Histology, Treatment Guide
Comprehensive pathology of dermoid cysts: from clinical presentation to histological diagnosis and management.
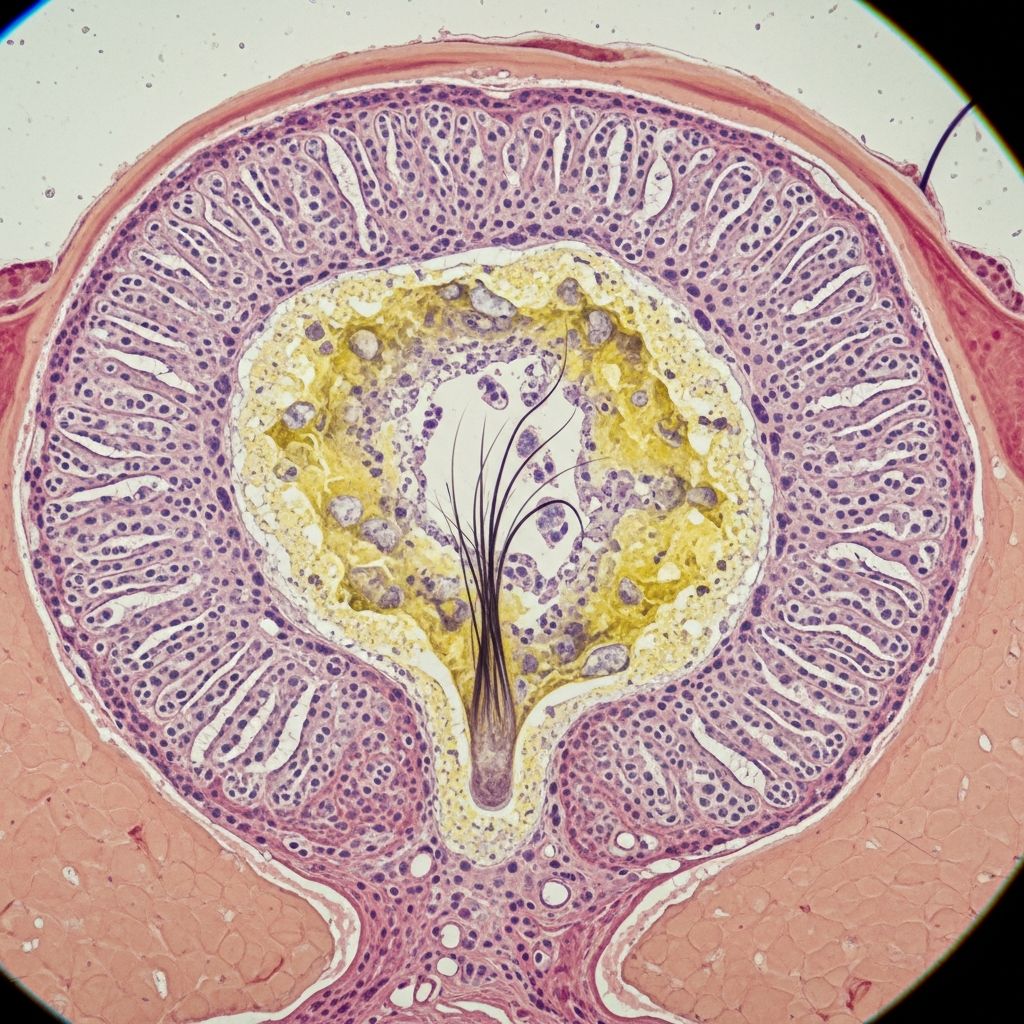
Dermoid Cyst Pathology
Dermoid cysts are benign congenital lesions classified as choristomas, consisting of normal tissues from germ cell layers ectopic to the site of presentation. These cysts arise from abnormal sequestration of epithelial cells during embryonic development, particularly along lines of fusion of embryonic tissue layers. In cutaneous pathology, they are most commonly subcutaneous and feature adnexal structures derived from ectoderm and mesoderm.
Introduction
Dermoid cysts represent a developmental anomaly where pluripotential cells from all three germ layers—ectoderm, mesoderm, and endoderm—can differentiate into mature tissues foreign to the anatomical location. Cutaneous dermoid cysts primarily involve ectodermal and mesodermal elements, such as skin, hair follicles, sebaceous glands, and sweat glands. They are distinct from teratomas, which are true neoplasms with disorganized tissue from all germ layers, whereas dermoid cysts exhibit organized, hamartomatous growth. These lesions are typically slow-growing, asymptomatic, and discovered incidentally or due to cosmetic concerns. Pathologically, they are submitted as ‘shelled out’ subcutaneous cysts, highlighting their well-circumscribed nature.
Embryologically, dermoid cysts form due to inclusion of epithelial rests along closure lines of the neural tube, branchial arches, or somites. In the skin, they often occur at fusion planes such as the supraorbital ridge or nasopalpebral groove. Unlike epidermoid cysts, which derive solely from surface epithelium, dermoid cysts contain adnexal appendages, making their pathology distinctive.
Clinical Features
Cutaneous dermoid cysts present as firm, nontender, doughy subcutaneous nodules, typically solitary and ranging from 0.5 to 6 cm in diameter. They are congenital, with 40% evident at birth and most manifesting by age 5. Common sites include the head and neck, particularly the lateral eyebrows (70% of cases), upper nasal bridge, and scalp. Less frequently, they appear on the trunk or extremities.
A key clinical clue is association with a central punctum or sinus tract in up to 45% of cases, which may drain cheesy keratinaceous material. The overlying skin is usually intact and mobile, though transillumination is negative due to solid contents like hair and keratin. Complications arise if the cyst ruptures, leading to inflammation, infection, or granulomatous reaction. Rarely, deeper extension into bone or orbit prompts neuroimaging.
- Firm, rounded subcutaneous mass
- Often midline or paramidline on head/neck
- Present at birth or early childhood
- May have overlying pit/sinus
- Slow growth; rarely symptomatic unless infected
Diagnosis
Diagnosis is primarily clinical, supported by characteristic location and appearance. Ultrasound reveals a well-defined hypoechoic mass with heterogeneous internal echoes from keratin, hair, and debris, aiding assessment of depth and sinus tracts. Fine-needle aspiration may yield keratin flakes but risks inflammation.
Definitive diagnosis requires histopathology post-excision. Imaging like CT or MRI is reserved for suspected intracranial/orbital extension, showing a non-enhancing cystic lesion with calcifications if teeth/dental structures are present (rare in cutaneous variants). Incisional biopsy is avoided due to recurrence risk from incomplete removal.
Histopathology
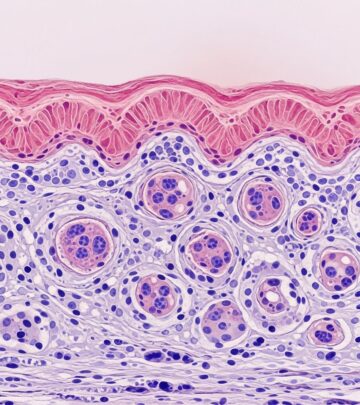
Microscopic examination reveals a unilocular or multilocular cyst in subcutaneous tissue, often fragmented from shelling out. The cyst wall is lined by stratified keratinizing squamous epithelium resembling epidermis, complete with a granular layer. The hallmark is adnexal differentiation: pilosebaceous units (hair follicles and sebaceous glands) attach directly to the cyst lining, with hair shafts freely floating in the lumen amid laminated keratin and sebaceous material.
Contents include desquamated keratin, cholesterol clefts, and inflammatory cells if ruptured. Less common findings encompass eccrine ducts, apocrine glands, smooth muscle bundles, or neural elements. Rarely, endodermal derivatives like respiratory or gastrointestinal epithelium appear. No atypia or mitoses indicate benignity.
- Cyst lining: Keratinizing stratified squamous epithelium with granular layer
- Adnexa: Hair follicles, sebaceous glands, sweat glands in wall
- Lumen: Hair shafts, keratin flakes, lipid debris
- Wall: Fibrous; may have smooth muscle or ectopic glands
Immunohistochemistry is rarely needed but shows CK5/6 positivity in squamous lining and adnexal markers like CEA for ducts.
Differential Diagnoses
Dermoid cysts must be distinguished from other subcutaneous cysts via clinical and histologic features. A table summarizes key differentials:
| Lesion | Key Features | Histology Distinguishes |
|---|---|---|
| Epidermoid cyst | No punctum; keratin only | Lacks adnexa/pilosebaceous units |
| Trichilemmal (pilar) cyst | Scalp; abrupt keratinization | Trichilemmal keratinization, no granular layer |
| Steatocystoma | Multiple; sebaceous | Sebaceous lobules connect to wavy, corrugated lining; no pilar structures |
| Dermoid sinus | Tract to dura | Communicating tract with glial heterotopia |
| Branchial cleft cyst | Lateral neck | Lymphoid tissue, squamous/columnar lining |
Other considerations include lipoma (soft, no punctum), pilomatrixoma (calcified, shadow cells), or rarely, basal cell carcinoma arising in a cyst.
Treatment
Surgical excision is curative, performed under general anesthesia in children, with complete removal of cyst wall and sinus tract to prevent recurrence (rate <5%). Approach varies by site: elliptical incision over the mass, careful dissection to preserve adjacent structures like orbicularis oculi. Intraoperative frozen section confirms margins if malignancy suspected.
Observation suffices for small, asymptomatic lesions. Infected cysts require incision/drainage and antibiotics first, followed by delayed excision. Minimally invasive options like laser ablation lack evidence for long-term efficacy. Histologic review of the specimen is mandatory to exclude rare squamous cell carcinoma (0.1-1%).
Complications and Prognosis
Untreated, dermoid cysts remain stable but risk infection (20%), rupture, or cosmetic deformity. Surgical complications include scarring, infection (5%), recurrence if incomplete (3-10%), or neural/vascular injury in head/neck sites. Malignancy is exceedingly rare, mostly in longstanding or ovarian dermoids.
Prognosis post-excision is excellent, with no malignant potential in properly managed cutaneous cases. Follow-up monitors for recurrence via clinical exam.
Frequently Asked Questions (FAQs)
Are dermoid cysts cancerous?
No, cutaneous dermoid cysts are benign. Malignant transformation to squamous cell carcinoma is extremely rare (<1%), warranting histologic exam post-excision.
Can dermoid cysts be removed without surgery?
Surgery is the gold standard. Aspiration or incision risks recurrence; observation is viable for asymptomatic cases.
Do dermoid cysts grow?
They grow slowly over years, typically stabilizing after childhood. Rapid enlargement suggests infection or hemorrhage.
Is a dermoid cyst the same as an epidermoid cyst?
No. Dermoids contain adnexal structures like hair follicles; epidermoids have only epidermal keratin.
When should imaging be done for a dermoid cyst?
Ultrasound routinely; MRI/CT if deep extension suspected, e.g., orbital or intracranial involvement.
Expanded Discussion on Pathophysiology
To delve deeper, dermoid cyst pathogenesis traces to 3rd-8th week embryogenesis. Epithelial entrapment at fusion lines (e.g., frontonasal prominence) leads to cystic dilation. Genetic syndromes like Goldenhar or Treacher Collins associate with complex dermoids, but isolated cases predominate.
Histologic variants include ‘hairy’ dermoids with prominent follicles versus ‘sebaceous’ types rich in glands. Inflammation elicits foreign body reaction to hair/keratin, mimicking abscess. Chronic cases fibrose the wall.
Comparative pathology underscores uniqueness: epidermoid cysts lack granular layer depth and adnexa; steatocystomas feature undulating epithelium without pilar differentiation.
In pediatric dermatopathology, distinguishing from nasal gliomas (solid, S100+) or encephalocele (brain tissue) is critical, often necessitating multidisciplinary input.
Surgical pearls include methylene blue sinogram for tracts and cautery of wall base to ablate microscopic residues. Cosmetic outcomes optimize via fusiform excision and layered closure.
Research gaps persist in molecular drivers; Wnt/APC pathway aberrations link to syndromic cysts akin to Gardner syndrome epidermoids. Future therapies may target cyst growth factors.
Epidemiology shows slight female predominance (1.5:1), possibly hormonal. Racial distribution mirrors population, ubiquitous globally.
References
- Epidermoid cyst – DermNet — DermNet NZ. 2023. https://dermnetnz.org/topics/epidermoid-cyst
- Epidermal Inclusion Cyst – StatPearls — NCBI Bookshelf, NIH. 2023-10-15. https://www.ncbi.nlm.nih.gov/books/NBK532310/
- Dermoid cyst pathology – DermNet — DermNet NZ. 2023. https://dermnetnz.org/topics/dermoid-cyst-pathology
- Dermoid cysts – DermNet — DermNet NZ. 2023. https://dermnetnz.org/topics/dermoid-cyst
- Cutaneous cysts and pseudocysts – DermNet — DermNet NZ. 2023. https://dermnetnz.org/topics/cutaneous-cysts-and-pseudocysts
Read full bio of medha deb