Graft-Versus-Host Disease Pathology: Essential Histology Guide
Detailed pathology of acute and chronic graft-versus-host disease affecting skin, GI tract, liver, and more post-HSCT.
Graft-versus-host disease pathology
Graft-versus-host disease (GVHD) is a potentially severe complication following allogeneic haematopoietic stem cell transplantation (HSCT), where donor immune cells attack the recipient’s tissues. This article examines the histopathological features of acute and chronic GVHD across affected organs, including skin, gastrointestinal tract, liver, and others, providing essential insights for diagnosis and management.
Introduction
Graft-versus-host disease arises when donor T lymphocytes recognize the recipient’s tissues as foreign, triggering an immune-mediated attack. Most commonly observed after allogeneic HSCT for haematological malignancies or immune disorders, GVHD affects multiple systems, with the skin being the earliest and most frequent site. Pathologically, it manifests in acute (typically within 100 days post-transplant) and chronic forms (beyond 100 days), each with distinct histological patterns.
The pathogenesis involves three phases: tissue damage from conditioning regimens releasing cytokines, activation of donor T cells by antigen-presenting cells, and effector phase with cytokine storm and organ infiltration. Incidence ranges from 40-60% post-HSCT, influenced by HLA mismatch, donor-recipient age disparity, and stem cell source.
Who is at risk of graft-versus-host disease?
Patients undergoing allogeneic HSCT are primarily at risk, particularly those with haematological cancers like leukaemia or lymphoma. Risk escalates with:
- HLA mismatch between donor and recipient
- Older recipient age
- Female donor to male recipient (due to minor histocompatibility antigens)
- Use of peripheral blood stem cells over bone marrow
- Myeloablative conditioning regimens
- Multiparity in female donors
Rarely, GVHD occurs post-solid organ transplant (e.g., liver), non-irradiated blood transfusion, or autologous HSCT.
Pathophysiology
GVHD pathophysiology unfolds in phases. Pre-transplant conditioning (chemotherapy/radiotherapy) damages host tissues, releasing danger signals like IL-1, TNF-α, and LPS, activating innate immunity via toll-like receptors. Host and donor antigen-presenting cells prime alloreactive donor T cells (Th1, Th17 subtypes), leading to proliferation and cytokine release (IFN-γ, IL-6). Effector cells infiltrate target organs, causing apoptosis and inflammation. Notably, GVHD confers graft-versus-leukemia effect, reducing relapse risk.
Histopathology of acute graft-versus-host disease
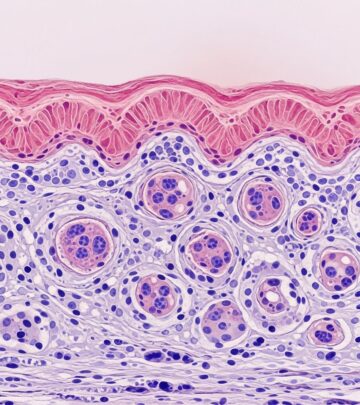
Acute GVHD (aGVHD) histopathology shows lichenoid interface dermatitis with lymphocyte exocytosis into the epidermis. Key features include:
- Skin: Basal vacuolar degeneration, dyskeratotic keratinocytes (Grade 1-2); subepidermal clefting, spongiosis (Grade 3); full-thickness epidermal necrosis (Grade 4).
| Grade | Skin Features |
|---|---|
| 1 | Focal/diffuse basal vacuolization |
| 2 | Basal vacuolization + spongiosis, dyskeratosis |
| 3 | Subepidermal cleft + dyskeratosis/spongiosis |
| 4 | Complete epidermal loss |
- Gut: Apoptotic bodies in crypts, glandular atrophy, dilated crypts; severe cases show crypt loss, surface denudation.
- Liver: Bile duct damage (dysplasia, dropout), cholestasis, endothelialitis; minimal portal inflammation.
Grading is organ-specific, with global scores predicting severity.
Histopathology of chronic graft-versus-host disease
Chronic GVHD (cGVHD) mimics autoimmune diseases, with sclerotic, lichenoid, or inflammatory patterns.
- Skin:
- Lichenoid: Dense dermal lymphocytic infiltrate, interface dermatitis.
- Sclerotic: Fibrosis in dermis/panniculus, resembling morphea; follicular mucinosis.
- Atrophic: Loss of adnexa, epidermal thinning.
- Gut: Subepithelial fibrosis (lamina propria), hyalinized stroma; less apoptosis than aGVHD.
- Liver: Chronic non-suppurative destructive cholangitis, bile duct loss, periductal fibrosis (like primary biliary cholangitis).
Overlap syndromes combine acute and chronic features.
Histopathology by organ
Skin
Skin involvement is universal in GVHD. Acute: Maculopapular rash evolving to erythroderma, bullae. Chronic: Poikiloderma, sclerosis, ulcers. Biopsy reveals satellite cell necrosis (isolated apoptotic keratinocytes rimmed by lymphocytes).
Gastrointestinal tract
Upper GI: Mucosal sloughing, apoptosis in gastric mucosa. Lower GI: Crypt cell necrosis, Paneth cell loss; severe: Total denudation. Fibrosis in chronic form impairs absorption.
Liver
Primarily bile duct epithelium targeted: vacuolization, attenuation, regenerative atypia. Portal triad expansion minimal; cholestasis prominent.
Lung
Bronchiolitis obliterans in cGVHD: Submucosal fibrosis, airway narrowing.
Other organs
Musculoskeletal: Fasciitis, myositis. Genital: Lichen sclerosus-like changes. Ocular: Conjunctival scarring.
Investigations
Diagnosis combines clinical (Glucksberg/NIH criteria), biopsy, and exclusion of infection/drug reactions. Skin biopsy is most accessible. Endoscopy/biopsy for GI; liver biopsy if needed. Molecular tests (e.g., TCR sequencing) emerging.
Differential diagnosis
- Acute skin: Drug eruption, viral exanthem, chemotherapy toxicity.
- Chronic skin: Scleroderma, lichen planus, morphea.
- GI: Infection (CMV), mucositis.
- Liver: Drug-induced cholestasis, viral hepatitis.
Histology distinguishes: GVHD lacks viral inclusions, has apoptotic bodies without viral cytopathy.
Treatment
Prophylaxis: Calcineurin inhibitors (cyclosporine/tacrolimus) + methotrexate/PTCy. Acute: Corticosteroids (1-2 mg/kg prednisone); refractory: Ruxolitinib, ibrutinib, extracorporeal photopheresis. Chronic: Steroids + IMiDs (thalidomide), JAK inhibitors, tyrosinase kinase inhibitors. Skin-directed: Topical steroids, PUVA.
Outcome
aGVHD grades 1-2 often resolve; grades 3-4 mortality >80%. cGVHD: 50% respond to steroids; NRM 20-30%. Long-term: Impaired QoL from sclerosis, infections.
Frequently Asked Questions
What is the most common organ affected in GVHD?
Skin, presenting as rash in 80% of cases, often first sign.
How is GVHD graded histologically in skin?
Using 4-tier system from basal vacuolization to epidermal loss.
Can GVHD occur after autologous transplant?
Rarely, yes, due to dysregulated immunity or minor antigens.
What is the role of HLA matching?
Mismatched HLA increases risk 2-5 fold.
Is mild GVHD beneficial?
Yes, associated with graft-versus-leukemia effect, lowering relapse.
References
- Graft-versus-host Disease — Penn Dermatology, University of Pennsylvania. 2023. https://dermatology.upenn.edu/clinical-programs/graft-versus-host-disease/
- Graft Vs. Host Disease — MedDerm. 2024. https://medderm.net/graft-versus-host-disease/
- Cutaneous Graft-Versus-Host Disease: Diagnosis and Treatment — NIH/PMC. 2018-01-15. https://pmc.ncbi.nlm.nih.gov/articles/PMC5797560/
- Graft-Versus-Host Disease — NCBI StatPearls. 2023-10-01. https://www.ncbi.nlm.nih.gov/books/NBK538235/
- Graft Versus Host Disease (GvHD) — DermNet NZ. 2024. https://dermnetnz.org/topics/graft-versus-host-disease
Read full bio of Sneha Tete