Impetigo Pathology: Clinical, Histologic & Pathogenic Insights
Detailed histopathological examination and clinical insights into impetigo, a common superficial bacterial skin infection.
Impetigo Pathology
Impetigo is a highly contagious superficial bacterial skin infection primarily affecting the epidermis. It is most common in children and typically caused by Staphylococcus aureus or Streptococcus pyogenes. This article delves into its clinical presentation, histopathological features, and pathological mechanisms across nonbullous and bullous variants.
Introduction
Impetigo represents one of the most frequent bacterial infections of the skin, characterized by erythematous plaques with yellow crusts that are itchy or painful. The infection spreads easily through direct contact, self-inoculation, or fomites, particularly in settings like schools or daycare centers. Nonbullous impetigo, the more prevalent form, accounts for about 70% of cases, while bullous impetigo is less common and toxin-mediated.
Pathologically, impetigo involves the stratum corneum and upper epidermis. Understanding its histopathology is crucial for distinguishing it from mimics and guiding therapy, especially in atypical or refractory cases.
Clinical Features
Impetigo begins with small vesicles or pustules that rupture rapidly, releasing purulent exudate that dries into the hallmark honey-colored crusts over an erythematous base. Lesions favor exposed areas like the face (perioral and perinasal regions), extremities, and sites of minor trauma. Satellite lesions form via autoinoculation, and mild regional lymphadenopathy may occur without systemic fever in uncomplicated cases.
Nonbullous Impetigo
The classic presentation involves multiple coalescing vesicles progressing to erosions covered by thick, golden-yellow crusts. Pruritus leads to scratching, exacerbating spread. Lesions heal without scarring in most cases but can pigment or evolve into ecthyma if deeper.
Bullous Impetigo
This variant features flaccid, fluid-filled bullae (1-2 cm) on intact skin, often in intertriginous areas, trunk, or extremities of infants. Bullae rupture leaves a collarette of scale without crusting. Fewer lesions occur, but systemic symptoms like fever may appear.
Ecthyma
A deeper, ulcerative form, ecthyma penetrates the dermis, forming punched-out ulcers with thick crusts and potential scarring. It arises from untreated impetigo or in immunocompromised hosts.
Histopathology
Diagnosis is usually clinical, but biopsy reveals distinctive features. Skin swabs or biopsies are reserved for atypical presentations, treatment failures, or outbreaks suspecting MRSA.
Nonbullous Impetigo
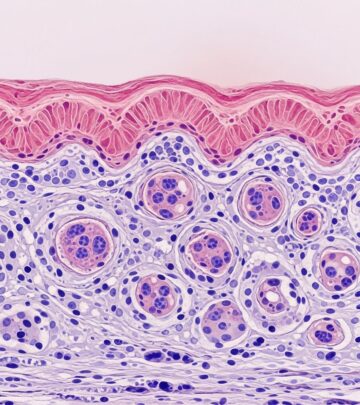
Microscopically, there is intra- or subcorneal acantholysis with neutrophils forming microabscesses in the stratum corneum. Gram-positive cocci (staphylococci or streptococci) cluster within these abscesses. The epidermis shows spongiosis, exocytosis, and a mixed inflammatory infiltrate of neutrophils and lymphocytes in the dermis. Crusts comprise degenerate stratum corneum, inflammatory cells, and bacteria.
- Key features: Subcorneal pustules, gram-positive bacteria, minimal dermal involvement.
- Surface: Honey-colored crust with bacterial colonies.
- Epidermis: Acanthosis, spongiosis.
- Dermis: Perivascular lymphocytic infiltrate with neutrophils.
Bullous Impetigo
Histology shows characteristic subcorneal clefting or bullae filled with neutrophils and acantholytic keratinocytes. Exfoliative toxins from phage group II S. aureus cleave desmoglein 1, causing intraepidermal split at the granular layer. Few organisms are seen, distinguishing it from nonbullous forms. Dermal edema and mild mixed infiltrate may be present.
- Diagnostic hallmarks: Large subcorneal bulla, sparse bacteria, acantholytic cells.
- No true crust formation post-rupture.
- Immunofluorescence negative, ruling out autoimmune bullous diseases.
Special Stains and Microbiology
Gram stain highlights cocci in clusters (staph) or chains (strep). Culture from pus confirms pathogen and sensitivities. PCR detects toxin genes in bullous cases. PAS or GMS stains exclude fungal mimics.
Pathogenesis
Bacteria colonize disrupted skin barriers (cuts, insect bites, eczema). Virulence factors include exfoliative toxins (ETA/ETB) for bullous impetigo, proteases, and superantigens. S. pyogenes produces streptolysin and hyaluronidase aiding spread. Host immunity limits depth, but poor hygiene or crowding promotes outbreaks.
In nonbullous impetigo, bacterial exotoxins and enzymes degrade desmosomes, leading to superficial splits. Immune response involves IgA antibodies, but ASO titers are unreliable for diagnosis.
Differential Diagnosis
| Condition | Key Distinguishing Features |
|---|---|
| Atopic dermatitis | Chronic, pruritic plaques without crusts; personal/family atopy history. |
| Herpes simplex | Grouped vesicles on erythematous base; Tzanck smear shows multinucleated giants. |
| Contact dermatitis | Geometric distribution; resolves with avoidance. |
| Scabies | Burrows, nocturnal itch; involves webs/folds. |
| Candidiasis | Satellite pustules in moist areas; KOH positive. |
| Varicella | Generalized polymorphic lesions; systemic symptoms. |
| Linear IgA bullous disease | Annular blisters; immunofluorescence shows linear IgA. |
Complications
Most cases self-limit in 2-3 weeks, but delays risk deeper infection. Post-streptococcal glomerulonephritis (PSGN) occurs 2-3 weeks post-infection in 1-2% (nephritic syndrome, ASO elevation). Scarring rare except ecthyma; pigmentation changes common. Neonates risk sepsis/meningitis. MRSA increases cellulitis risk.
- PSGN: Hypocomplementemic, resolves in most but monitor renal function.
- Rheumatic fever: Rare with skin strains.
- Lymphangitis or abscess: If untreated.
Management
Topical mupirocin or fusidic acid for localized disease; oral cephalexin, dicloxacillin, or clindamycin for extensive/MRSA cases. Duration: 5-7 days. Hygiene: Remove crusts with soap/water, avoid sharing items. Isolate until non-contagious (24h antibiotics or crusted).
Supportive: Antihistamines for itch, incision/drainage for ecthyma.
Prognosis
Excellent with treatment; resolution in 7-10 days. Untreated: 2-3 weeks. Recurrence common in at-risk groups.
Frequently Asked Questions
What causes impetigo?
Primarily S. aureus (80%) and S. pyogenes (20%), entering via skin breaks.
Is impetigo contagious?
Yes, via direct contact or fomites until crusted or 24h post-antibiotics.
How is impetigo diagnosed?
Clinically; swabs/biopsy if refractory or outbreak.
Does impetigo scar?
Rarely, except ecthyma form.
Can impetigo lead to kidney problems?
Yes, PSGN in streptococcal cases.
Prevention
- Maintain skin integrity; treat underlying dermatitis.
- Hand hygiene, avoid sharing towels.
- Early treatment in outbreaks.
References
- Impetigo – StatPearls — NCBI Bookshelf. 2023. https://www.ncbi.nlm.nih.gov/books/NBK430974/
- Impetigo – Symptoms & causes — Mayo Clinic. 2023-10-10. https://www.mayoclinic.org/diseases-conditions/impetigo/symptoms-causes/syc-20352352
- Impetigo – Diagnosis & treatment — Mayo Clinic. 2023-10-10. https://www.mayoclinic.org/diseases-conditions/impetigo/diagnosis-treatment/drc-20352358
- Impetigo: Diagnosis and Treatment — AAFP. 2014-08-15. https://www.aafp.org/pubs/afp/issues/2014/0815/p229.html
- Impetigo, Contagious Skin Infection — Cleveland Clinic. 2023. https://my.clevelandclinic.org/health/diseases/15134-impetigo
- Impetigo – treatments, symptoms and causes — Healthdirect (Australian Government). 2023. https://www.healthdirect.gov.au/impetigo
Read full bio of medha deb