Infantile Haemangioma: Definition and Pathogenesis
Exploring the definition, causes, clinical evolution, and cellular origins of infantile haemangioma, the most common benign vascular tumour in infants.
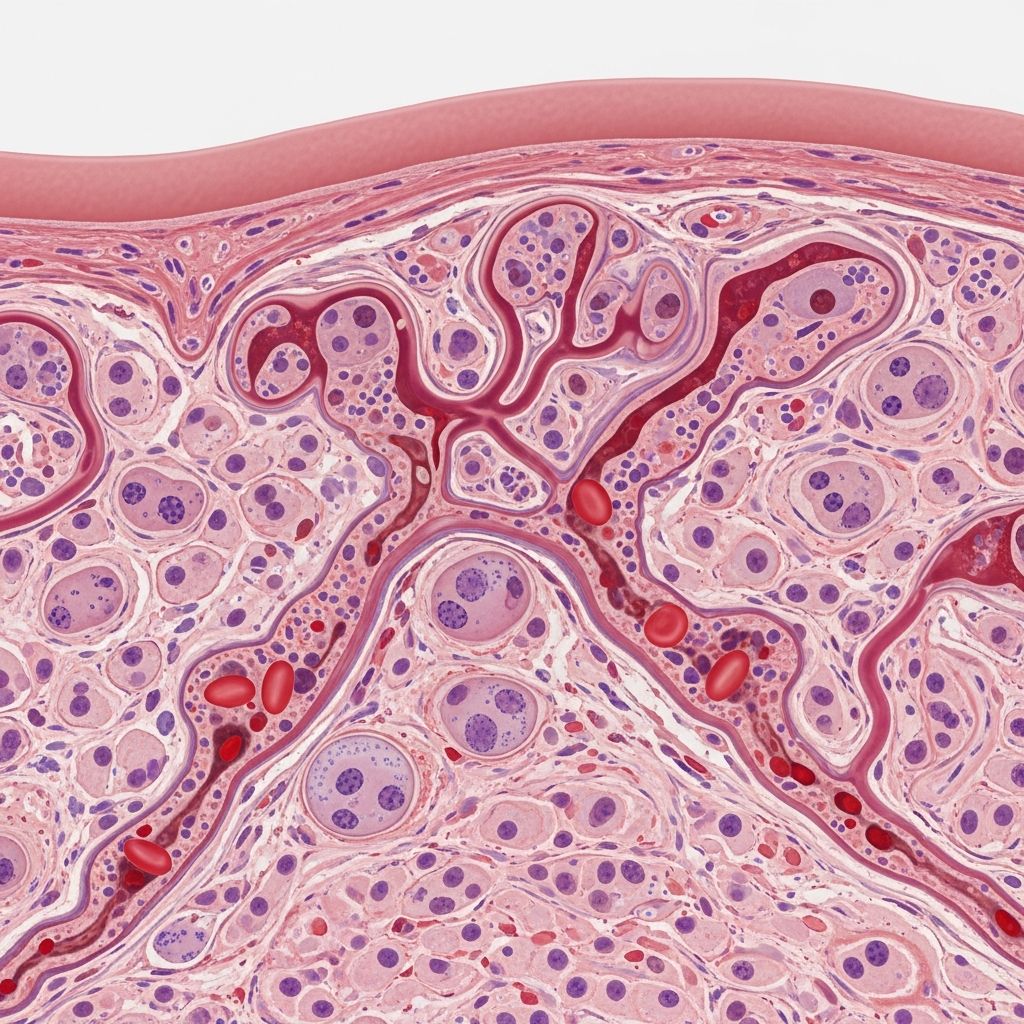
Infantile haemangioma, also known as a strawberry naevus, represents the most prevalent benign vascular skin tumour observed in children. These lesions typically become noticeable within the initial weeks following birth and follow a characteristic pattern of proliferation followed by spontaneous involution.
Introduction
Infantile haemangiomas (IH) are benign vascular neoplasms that arise from endothelial cell proliferation, distinguishing them from vascular malformations, which are congenital anomalies present at birth. Unlike malformations, IH exhibit a life cycle involving rapid postnatal growth and eventual regression, affecting approximately 5% of infants. They are classified based on depth as superficial (strawberry-like, elevated red lesions), deep (bluish subcutaneous nodules), or mixed. This unique behaviour underscores their distinct pathogenesis involving dysregulated angiogenesis and vasculogenesis.
Demographics
Infantile haemangiomas occur in 1–2% of newborns, with prevalence rising to 10–12% by one year of age among Caucasian infants. The incidence is lower in non-Caucasian populations. A striking female predominance exists, with a female-to-male ratio of 3:1. Prematurity, low birth weight (<1500 g), and multiple gestation pregnancies significantly elevate risk, as these factors correlate with higher hypoxic exposure in utero.
- Prevalence: 1–2% at birth, up to 12% at 1 year (Caucasian infants).
- Sex ratio: Females 3 times more affected than males.
- High-risk groups: Premature infants, multiples, low birth weight.
Causes
The aetiology of infantile haemangioma remains multifactorial, encompassing genetic predispositions, environmental triggers, and developmental anomalies. No single causative mutation has been consistently identified, suggesting epigenetic and extrinsic factors play pivotal roles. Key theories include placental trophoblast embolization, hypoxia-induced vasculogenesis, and dysregulation of vascular stem cells.
Risk factors
Several clinical associations heighten susceptibility:
- Prematurity and low birth weight.
- Female sex.
- Multiple births (twins or more).
- Maternal factors: advanced age, pre-eclampsia, chorionic villus sampling.
- White ethnicity.
These factors often link to intrauterine hypoxia, promoting endothelial progenitor cell mobilization.
Pathogenesis
IH pathogenesis centres on aberrant vasculogenesis (de novo vessel formation from progenitors) and angiogenesis (sprouting from existing vessels). Hypoxia emerges as a primary initiator, upregulating hypoxia-inducible factor (HIF-1α and HIF-2α), which induces vascular endothelial growth factor (VEGF) and glucose transporter 1 (GLUT1). GLUT1 expression serves as a diagnostic hallmark, absent in differentials like congenital haemangiomas.
Placental origin theory posits that trophoblastic stem cells embolize to fetal circulation, expressing fetal markers like CD133 from the cardinal vein. These cells resemble placental vasculature histologically and immunohistochemically. Endothelial progenitor cells (EPC) and hemangioma stem cells (HemSC) contribute to lobular capillary proliferation, supported by perivascular cells and pericytes.
The renin-angiotensin system may amplify proliferation, with elevated renin in young infants correlating with IH risk. Angiotensin II stimulates endothelial growth. Genetic studies reveal rare familial cases and associations with syndromes like PHACE, but most are sporadic, implicating epigenetic mechanisms such as SOX18 transcription factor dysregulation.
| Mechanism | Description | Key Markers/Mediators |
|---|---|---|
| Hypoxia | Initiates progenitor mobilization | HIF-1α, VEGF, GLUT1 |
| Placental Theory | Trophoblast embolization | CD133, fetal vein markers |
| Stem Cell Dysregulation | EPC and HemSC proliferation | CD31, CD133 |
| Angiogenesis | Vessel sprouting | VEGF, renin-angiotensin |
Clinical features
Typically solitary (80–90%), IH predominantly affect the head and neck (60%), followed by trunk and limbs. Multiple lesions (>5) suggest visceral involvement, warranting screening. Depth determines appearance: superficial (bright red, strawberry-like), deep (blue, subcutaneous), mixed. Distribution can be focal, segmental (risk for ulceration/PHACE), or indeterminate.
Depth
- Superficial: Dermal, raised, red, blanches with pressure.
- Deep: Subcutaneous, blue-purple, poorly defined.
- Mixed: Combined features.
Distribution
- Focal: Localized, round/oval (<5 cm).
- Segmental: Dermatomal, larger (>5 cm), higher complication risk.
- Indeterminate: Patchy, non-dermatomal.
Growth phases
IH evolve through predictable phases:
- Nascent/Premonitory (birth–2 weeks): Pale or telangiectatic patch, subtle precursor.
- Proliferation (2 weeks–5 months): Rapid expansion (up to 1 cm/month), thickening, may ulcerate (especially segmental).
- Plateau (5–10 months): Growth stabilizes.
- Involution (1–10 years): Greyish discoloration, softening, regression; 50% complete by age 5, 90% by 9 years. Residual telangiectasia or fat atrophy in 50%.
A subtype, infantile haemangioma with minimal or arrested growth (IH-MAG), shows <25% proliferation, mimicking port-wine stains, often on limbs with syndromic links.
Histology
Proliferative phase: Lobular capillary clusters with plump endothelial cells, mitotic figures, GLUT1+, CD31+, CD34+. Involuting phase: Vessel dilation, fibrosis, flattened endothelium. Multinodular pattern fed by lobular arterioles.
Diagnosis
Primarily clinical, based on age of onset, growth dynamics, and GLUT1 positivity on biopsy (if needed). Imaging (ultrasound/Doppler) assesses depth/vascularity; MRI for hepatic/multifocal cases. Differentials include congenital haemangioma (present at birth, GLUT1-), pyogenic granuloma, tufted angioma, kaposiform hemangioendothelioma.
| Feature | Infantile Haemangioma | Congenital Haemangioma | Port-Wine Stain |
|---|---|---|---|
| Onset | Postnatal weeks | At birth, fully grown | At birth, flat |
| Growth | Proliferates then involutes | Involutes or grows rapidly | Static/progressive |
| GLUT1 | Positive | Negative | Negative |
| Complications | Ulceration, obstruction | Heart failure (RICH) | Hypertrophy |
Frequently Asked Questions (FAQs)
Q: What is the most common location for infantile haemangioma?
A: Head and neck region (60% of cases), followed by trunk and extremities.
Q: Do all infantile haemangiomas regress completely?
A: Approximately 50% leave minor residuals like telangiectasia or atrophy; full clearance is less common.
Q: Is infantile haemangioma cancerous?
A: No, it is a benign tumour with no malignant potential.
Q: When should treatment be considered?
A: For functional impairment, ulceration, or high-risk sites (periorbital, airway); propranolol is first-line.
Q: Can infantile haemangiomas affect internal organs?
A: Yes, especially hepatic in multifocal cutaneous cases; screening recommended.
Understanding infantile haemangioma’s natural history reassures most families, as spontaneous resolution occurs, though vigilant monitoring prevents complications. Multidisciplinary care optimizes outcomes in complex cases.
References
- Infantile Haemangioma: Classification, Diagnosis, & Management — The Plastics Fella. 2023. https://www.theplasticsfella.com/infantile-haemangioma/
- Infantile haemangioma: Definition and pathogenesis — DermNet NZ. 2023-10-01. https://dermnetnz.org/topics/infantile-haemangioma-definition-and-pathogenesis
- Infantile hemangioma: the common and enigmatic vascular tumor — Journal of Clinical Investigation (JCI). 2022-06-01. https://www.jci.org/articles/view/172836
- Infantile Hemangiomas: An Update on Pathogenesis and Treatment — PubMed Central (PMC). 2021-10-19. https://pmc.ncbi.nlm.nih.gov/articles/PMC8539430/
- Infantile Hemangiomas — American Academy of Pediatrics (AAP). 2023. https://publications.aap.org/pediatriccare/article/doi/10.1542/aap.ppcqr.396482/150/Infantile-Hemangiomas
Read full bio of Sneha Tete