Invasive Ductal Carcinoma: Types, Diagnosis & Treatment
Comprehensive guide to invasive ductal carcinoma: understanding IDC breast cancer, diagnosis, and treatment options.
Understanding Invasive Ductal Carcinoma (IDC)
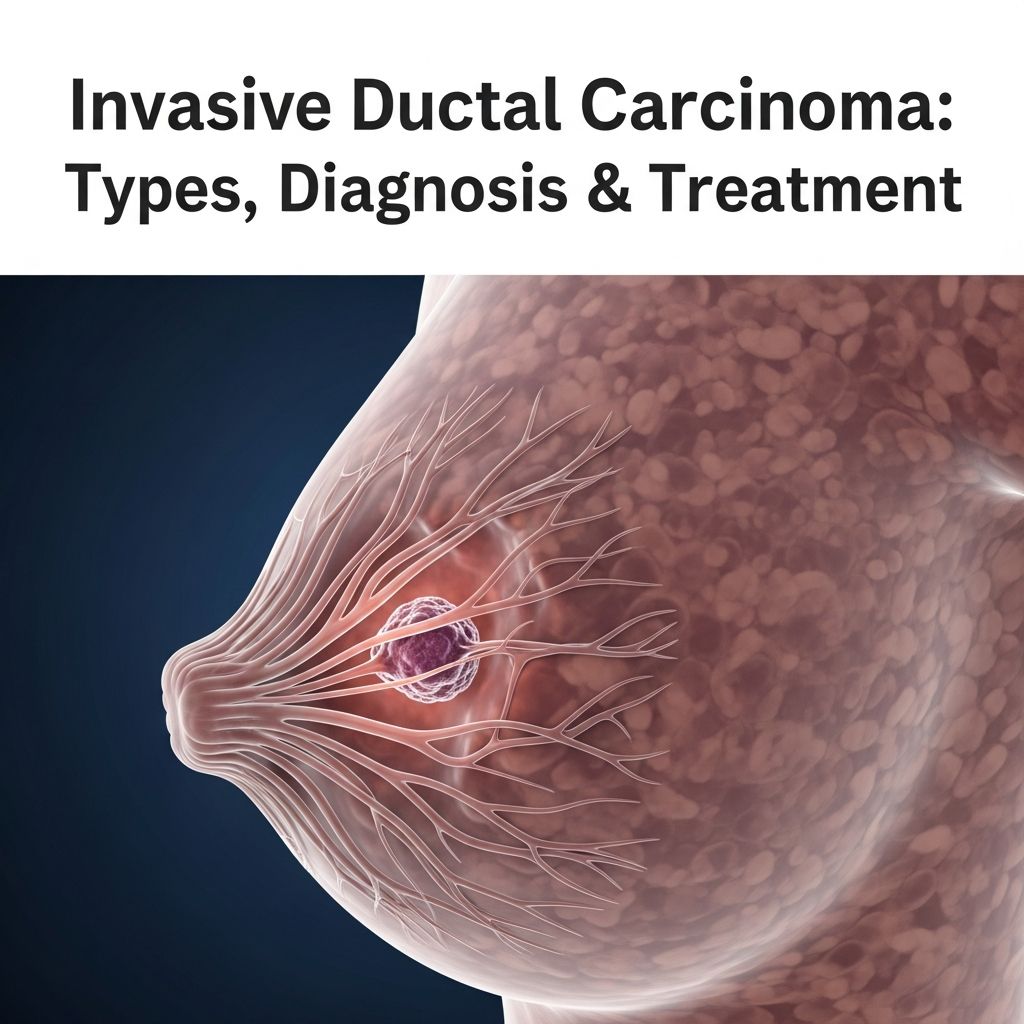
Invasive ductal carcinoma (IDC), also known as infiltrating ductal carcinoma, represents the most common form of breast cancer diagnosed in patients today. Accounting for approximately 80% of all breast cancer cases, IDC is a significant health concern affecting thousands of individuals each year. This type of cancer begins in the milk ducts of the breast—the tubes responsible for carrying milk from the milk-producing lobules to the nipple—and then spreads into the surrounding breast tissue. Understanding IDC is crucial for early detection, appropriate treatment planning, and improving patient outcomes.
The term “invasive” specifically refers to the cancer’s ability to break through the walls of the milk ducts and penetrate surrounding breast tissue. Unlike ductal carcinoma in situ (DCIS), which remains confined within the duct walls, IDC has the potential to spread to lymph nodes and other areas of the body through the lymphatic system or bloodstream, a process known as metastasis. This distinguishing characteristic makes IDC a more serious concern requiring prompt medical attention and comprehensive treatment strategies.
Key Characteristics of IDC
Several important features distinguish invasive ductal carcinoma from other breast cancer types:
- Origin in milk ducts: IDC begins in the epithelial cells lining the breast’s milk ducts
- Invasive nature: Cancer cells break through the basement membrane and invade surrounding breast tissue
- Metastatic potential: IDC can spread to lymph nodes, bones, lungs, liver, brain, and other distant organs
- Prevalence in males: IDC is also the most common type of male breast cancer
- Variable growth rates: IDC can grow slowly or aggressively depending on tumor grade and molecular characteristics
Symptoms and Detection
Many individuals with early-stage invasive ductal carcinoma may not experience any symptoms, which is why routine screening is essential. However, when symptoms do appear, they may include:
- A new, painless lump in the breast, either too small to feel or large enough to be palpable
- Swelling or thickening in part of the breast
- Skin dimpling or puckering
- Nipple retraction or inversion
- Nipple discharge, particularly if bloody or clear
- Redness or scaling of the breast skin
- Breast pain or discomfort in specific areas
- Suspicious calcifications detected on mammography
IDC is typically diagnosed following routine breast cancer screenings such as mammograms or clinical breast examinations. If a healthcare provider suspects breast cancer based on imaging or physical examination findings, additional diagnostic tests and a breast biopsy will be performed to confirm the diagnosis.
Diagnostic Procedures
When invasive ductal carcinoma is suspected, several diagnostic methods may be employed to confirm the diagnosis and gather information needed for treatment planning:
Mammography
Mammography remains the primary screening tool for detecting breast abnormalities. Digital mammography and 3D mammography (tomosynthesis) can help identify suspicious areas requiring further investigation. Calcifications or masses detected on mammography often prompt additional testing.
Ultrasound
Breast ultrasound uses sound waves to create detailed images of breast tissue. This imaging modality is particularly useful for distinguishing between solid tumors and fluid-filled cysts, helping determine whether a suspicious area requires biopsy.
Magnetic Resonance Imaging (MRI)
Breast MRI provides high-resolution images and may be recommended in specific clinical situations, such as evaluating the extent of disease or assessing the contralateral breast.
Biopsy
A biopsy is the definitive diagnostic procedure for confirming invasive ductal carcinoma. During a biopsy, a tissue sample is collected from the suspicious area using a needle or surgical technique. The tissue is then examined under a microscope by a pathologist to confirm the diagnosis and determine specific tumor characteristics.
Staging and Grading Systems
Once IDC is diagnosed, the cancer is staged and graded to guide treatment decisions and provide prognostic information. These systems help healthcare providers determine the extent of disease and predict treatment response.
IDC Staging
The TNM staging system (Tumor, Node, Metastasis) classifies IDC into stages 0-4 based on tumor size, lymph node involvement, and metastatic spread:
| Stage | Characteristics |
|---|---|
| Stage 1 | Cancer has spread from the milk duct into breast tissue but has not spread to lymph nodes or distant sites |
| Stage 2A | Tumor is 2-5 centimeters without lymph node involvement, or smaller than 2 centimeters with spread to nearby lymph nodes |
| Stage 2B | Tumor is larger than 5 centimeters without lymph node spread, or 2-5 centimeters with axillary lymph node involvement |
| Stage 3A | Cancer has spread to nearby lymph nodes or chest wall tissue |
| Stage 3B | Cancer has invaded the chest wall or breast skin, or inflammatory breast cancer characteristics present |
| Stage 3C | Cancer has spread to lymph nodes near the collarbone |
| Stage 4 | Cancer has metastasized to distant organs such as bones, liver, lungs, or brain (metastatic breast cancer) |
IDC Grading
Tumor grade reflects how much cancer cells resemble normal breast cells and predicts growth rate. The Nottingham Histologic Grade system assigns grades 1-3:
| Grade | Appearance & Behavior |
|---|---|
| Grade 1 (Low) | Cancer cells closely resemble normal breast cells and grow slowly |
| Grade 2 (Intermediate) | Cancer cells have moderate differentiation and grow at a moderate pace |
| Grade 3 (High) | Cancer cells look distinctly abnormal and grow rapidly; associated with higher recurrence risk |
Molecular Subtypes and Receptor Status
Beyond traditional staging and grading, IDC tumors are classified based on hormone and protein receptor status, which significantly influences treatment selection:
- Hormone Receptor-Positive (HR+): Approximately 69% of invasive breast cancers express estrogen or progesterone receptors, making them responsive to hormone-blocking therapies
- HER2-Positive: About 4% of invasive breast cancers overexpress the HER2 protein, requiring targeted HER2-directed therapy
- Triple-Negative: Approximately 15% of breast cancers lack estrogen, progesterone, and HER2 receptors; these tumors tend to grow more aggressively and require chemotherapy as primary treatment
Treatment Options for IDC
Treatment plans for invasive ductal carcinoma are individualized based on tumor characteristics, stage, receptor status, patient age, overall health, and personal preferences. Most patients receive multimodal therapy combining surgery, radiation, and/or systemic treatments.
Surgical Treatment
Lumpectomy (Breast-Conserving Surgery): This procedure removes the tumor and surrounding margin of normal tissue while preserving the breast. Lumpectomy is typically followed by radiation therapy to reduce recurrence risk and is often preferred for early-stage IDC when adequate margins can be achieved.
Mastectomy: Complete removal of the breast tissue may be recommended for larger tumors, multiple tumors, widespread disease, or when adequate margins cannot be achieved with lumpectomy. Mastectomy may be unilateral or bilateral depending on cancer extent and patient preference.
Lymph Node Evaluation: Sentinel lymph node biopsy or axillary lymph node dissection may be performed to determine lymph node involvement, which is crucial for staging and treatment planning.
Radiation Therapy
Radiation therapy uses high-energy beams to destroy cancer cells and is frequently recommended after lumpectomy to eliminate any remaining cancer cells and reduce breast cancer recurrence risk. Radiation may also be used after mastectomy in specific circumstances, such as large tumors, lymph node involvement, or positive margins.
Systemic Therapies
Chemotherapy: Chemotherapy drugs kill rapidly dividing cancer cells throughout the body. Chemotherapy may be administered before surgery (neoadjuvant) to shrink tumors or after surgery (adjuvant) to eliminate microscopic metastatic disease. For triple-negative IDC, chemotherapy is the primary systemic treatment option.
Hormone Therapy: Patients with hormone receptor-positive tumors benefit from endocrine therapy using medications such as tamoxifen or aromatase inhibitors. These drugs block estrogen’s effects on cancer cells, reducing recurrence risk and improving survival outcomes.
Targeted Therapy: HER2-positive tumors respond to targeted monoclonal antibodies and tyrosine kinase inhibitors such as trastuzumab (Herceptin) or pertuzumab, which specifically target HER2-driven tumor growth.
Immunotherapy: For certain patients, particularly those with triple-negative breast cancer, immunotherapy medications that activate the immune system to recognize and destroy cancer cells may be combined with chemotherapy.
Prognosis and Survival Rates
When discovered and treated early, the five-year survival rate for invasive ductal carcinoma is nearly 100 percent. However, survival rates vary significantly based on cancer stage at diagnosis:
- Early-stage disease: Five-year survival rates exceed 90% for stages 1-2
- Locally advanced disease: Five-year survival rates decrease to 40-65% for stage 3
- Metastatic disease: Five-year survival rates for stage 4 disease are lower, though improving with newer treatment options
Factors influencing prognosis include tumor size, grade, lymph node involvement, hormone receptor status, HER2 status, patient age, and overall health. Ongoing research and development of novel therapies continue to improve outcomes for patients with advanced invasive ductal carcinoma.
Risk Factors and Prevention
While the exact cause of IDC remains unknown, several risk factors increase the likelihood of developing this cancer:
- Age (risk increases with advancing age)
- Female sex
- Personal history of breast cancer
- Family history of breast cancer
- Genetic mutations (BRCA1, BRCA2)
- Estrogen and progesterone hormone exposure
- Obesity
- Alcohol consumption
- Radiation exposure
- Dense breast tissue
While not all risk factors are modifiable, maintaining a healthy weight, limiting alcohol consumption, regular exercise, and hormone therapy decisions can help reduce breast cancer risk.
Follow-Up Care and Survivorship
After completing primary treatment for IDC, regular follow-up care is essential to monitor for recurrence and manage treatment side effects. Survivorship care includes:
- Regular clinical breast examinations
- Imaging surveillance with mammography or MRI
- Management of treatment-related side effects
- Psychological support and counseling
- Lifestyle modification and wellness programs
- Genetic counseling when indicated
Frequently Asked Questions
Q: What is the difference between IDC and DCIS?
A: Ductal carcinoma in situ (DCIS) is non-invasive cancer confined within the milk duct that has not spread to surrounding tissue. IDC is invasive cancer that has penetrated beyond the duct into surrounding breast tissue and has potential to metastasize.
Q: Can IDC be cured?
A: When caught early and treated appropriately, IDC can be cured. Early-stage disease has nearly 100% five-year survival rates. Advanced disease is more difficult to cure but increasingly manageable with newer treatment options.
Q: Is IDC hereditary?
A: While most IDC cases are sporadic, inherited genetic mutations such as BRCA1 and BRCA2 significantly increase breast cancer risk. Genetic counseling and testing may be recommended for individuals with strong family histories.
Q: How often should I have breast cancer screening?
A: Screening recommendations depend on age and risk factors. Generally, women ages 40-44 may begin screening, and women 45-54 should receive annual mammograms. Those with higher risk factors may require more frequent screening or supplemental imaging.
Q: What should I expect during treatment?
A: Treatment experiences vary based on therapy type. Surgery involves recovery time, radiation requires multiple outpatient visits, and chemotherapy may cause fatigue and side effects. Your healthcare team will provide detailed information about what to expect.
Q: Can IDC recur after treatment?
A: Yes, IDC can recur locally in the breast or at distant sites. Regular follow-up care, imaging surveillance, and adherence to recommended therapies help detect recurrence early when treatment options remain available.
References
- Invasive Ductal Carcinoma (IDC) Breast Cancer — Penn Medicine. 2024. https://www.pennmedicine.org/conditions/invasive-ductal-carcinoma
- Invasive Ductal Carcinoma (IDC): Symptoms, Treatments, and More — Breast Cancer Organization. 2024. https://www.breastcancer.org/types/invasive-ductal-carcinoma
- Invasive Ductal Carcinoma (IDC): Overview, Treatment & Prognosis — National Breast Cancer Foundation. 2024. https://www.nationalbreastcancer.org/invasive-ductal-carcinoma/
- Invasive Ductal Carcinoma (IDC) — University of Michigan Health. 2024. https://www.uofmhealth.org/our-care/specialties-services/breast-cancer/invasive-ductal-carcinoma-idc
- Invasive Ductal Carcinoma (IDC) Breast Cancer — Breast Cancer Research Foundation. 2024. https://www.bcrf.org/about-breast-cancer/invasive-ductal-carcinoma/
- Invasive Ductal Carcinoma (IDC): Stages & Treatment — Cleveland Clinic. 2024. https://my.clevelandclinic.org/health/diseases/22117-invasive-ductal-carcinoma-idc
- Invasive Ductal Carcinoma Stages, Treatment and Survival Rate — Cancer Center. 2024. https://www.cancercenter.com/cancer-types/breast-cancer/types/common-breast-cancer-types/invasive-ductal-carcinoma
Read full bio of Sneha Tete