Lichenoid Drug Reaction Pathology: 7 Key Histologic Features
Comprehensive pathological analysis of lichenoid drug reactions, including histology, clinical features, and differential diagnosis.
Lichenoid Drug Reaction Pathology
Authoritative facts
Lichenoid drug reactions (LDRs), also known as drug-induced lichenoid eruptions, represent a distinctive subset of adverse cutaneous drug reactions mimicking idiopathic lichen planus (LP). These hypersensitivity reactions arise from medications altering epidermal antigens, triggering a T-cell-mediated interface dermatitis. Unlike classic LP, LDRs often exhibit atypical clinical distributions, prolonged latency periods, and specific histological clues such as parakeratosis and eosinophils.
Histopathologically, LDRs feature a lichenoid lymphocytic infiltrate at the dermoepidermal junction with basal keratinocyte vacuolar degeneration. The mean onset latency is approximately 15.7 weeks post-drug initiation, ranging from days to years, with resolution typically occurring over 14.2 weeks after drug cessation. Checkpoint inhibitors, tyrosine kinase inhibitors, and biologics are leading culprits in contemporary cases.
Microscopic (histopathologic) description
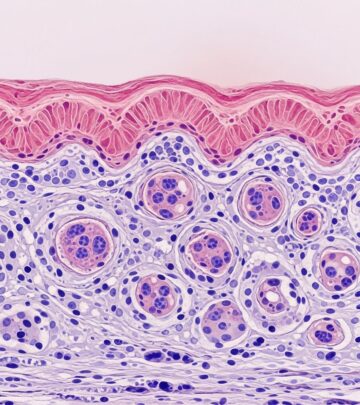
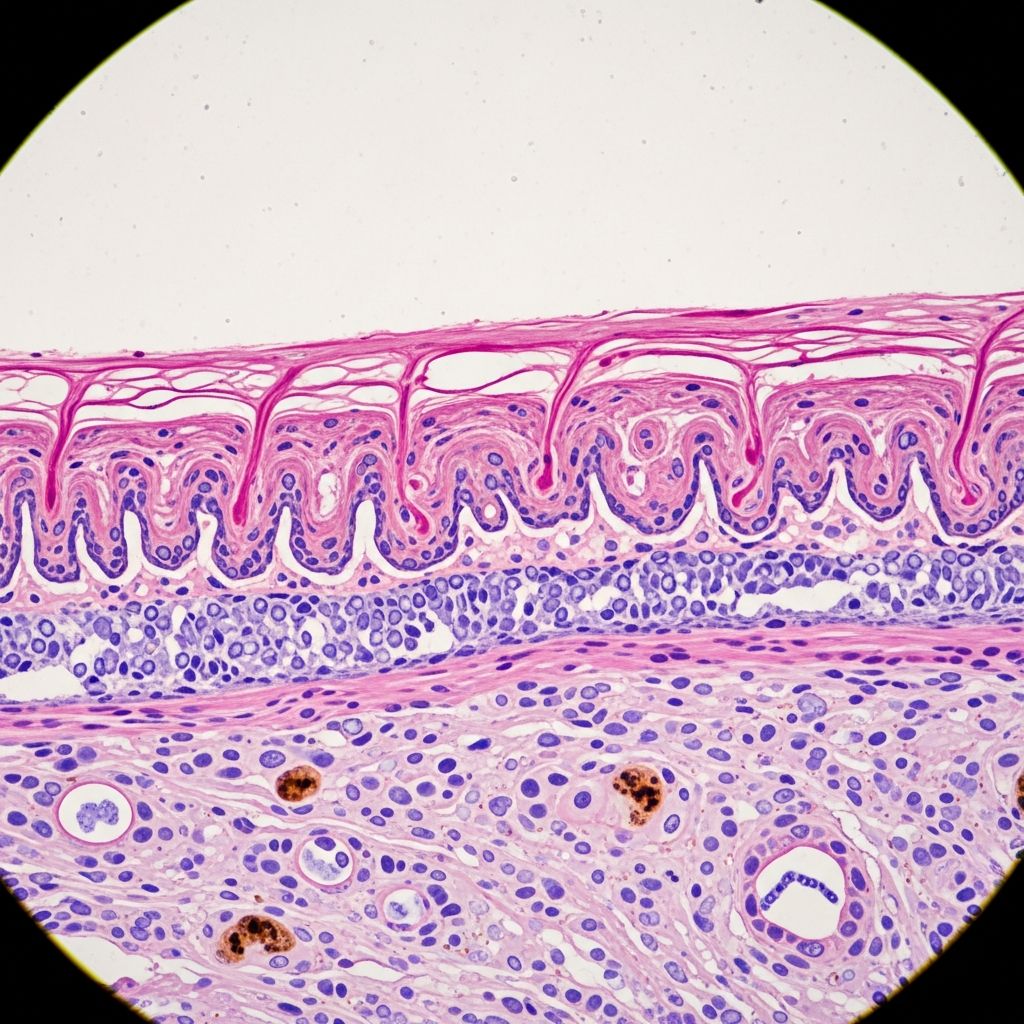
The hallmark of lichenoid drug reaction pathology is interface dermatitis characterized by a band-like lymphocytic infiltrate obscuring the dermoepidermal junction. Key features include:
- Vacuolar degeneration of basal keratinocytes, forming a “saw-tooth” rete ridge appearance in mild cases.
- Civatte bodies (colloid or apoptotic keratinocytes) prominent in the lower epidermis and stratum corneum.
- Parakeratosis, often focal or confluent, distinguishing from classic LP which typically shows compact orthokeratosis.
- Eosinophils scattered within the infiltrate or dermis, a pivotal differentiator (absent or rare in idiopathic LP).
- Hypergranulosis with irregular acanthosis; wedge-shaped hypergranulosis may occur.
- Upper dermal oedema and dilated capillaries in acute phases.
- Mild to moderate fibrosis in chronic lesions, with vertical collagen stretching between rete ridges.
Variations include psoriasiform hyperplasia in subacute presentations and pigment incontinence leading to postinflammatory hyperpigmentation.
| Feature | Lichenoid Drug Reaction | Idiopathic Lichen Planus |
|---|---|---|
| Parakeratosis | Frequent (focal/confluent) | Rare |
| Eosinophils | Common (infiltrate/dermis) | Rare/absent |
| Interface infiltrate | Patchy/perivascular | Band-like, continuous |
| Civatte bodies | Numerous, superficial | Few, basal |
| Granular layer | Hyperplastic, irregular | Hyperplastic, uniform |
| Ornamentation | Preserved/variable | Lost (flattened rete) |
Positive stains
- CD4:CD8 ratio often inverted (CD8+ predominant), mirroring LP but with more CD4+ cells peripherally.
- CD68+ macrophages engulf apoptotic debris.
- Granzyme B, perforin expression in cytotoxic T-cells targeting keratinocytes.
- ICAM-1 upregulated on basal keratinocytes, facilitating leukocyte adhesion.
Negative stains
- Direct immunofluorescence (DIF): Typically negative or nonspecific; shaggy fibrinogen at basement membrane possible, but lacks true LP’s linear IgM/C3 “hydrophilic plug” pattern.
- PD-L1/PD-1: Variable in non-checkpoint inhibitor cases.
Pathophysiology and etiology
LDRs stem from haptenization where drugs bind keratinocytes, generating neoantigens. This activates CD8+ cytotoxic T-cells via perforin/granzyme-mediated apoptosis, amplified by IFN-γ from CD4+ helpers. Plasmacytoid dendritic cells contribute via IFN-α production.
Common culprit drugs (categorized by prevalence):
| Category | Examples | Frequency |
|---|---|---|
| Checkpoint inhibitors | PD-1/PD-L1 inhibitors (nivolumab, pembrolizumab) | 42.1% |
| Tyrosine kinase inhibitors | sunitinib, imatinib, erlotinib | 12.0% |
| Biologics | anti-TNF (infliximab, adalimumab) | 4.0% |
| Antimalarials | quinine, chloroquine | Classic |
| Antihypertensives | ACE inhibitors, β-blockers | Common |
| Diuretics | thiazides, furosemide | Frequent |
| NSAIDs | gold salts (historic), phenylbutazone | Variable |
Differential diagnosis
Distinguishing LDR from mimics requires clinicopathologic correlation:
- Idiopathic lichen planus: Uniform band-like infiltrate, no eosinophils/parakeratosis, oral/genital involvement common.
- Lichenoid contact dermatitis: Localized, neomycin/quaternium-15 exposure, spongiosis prominent.
- Graft-versus-host disease: Apoptotic keratinocytes sparse, satellite cell necrosis, post-transplant history.
- Lupus erythematosus: Thickened basement membrane, dermal mucin, positive DIF (IgG/IgM).
- Syphilis (secondary): Plasma cells abundant, endemic treponemal history.
- Pityriasis lichenoides: Interface changes with parakeratosis, but prominent exocytosis.
Clinical features
LDRs present as pruritic, violaceous papules/plaques with “violaceous” hue, often photosensitive distribution (face, upper trunk). Unlike LP, lesions may be widespread, annular, or show central clearing. Latency averages 1 year (range 1 week–several years); resolution lags 1–18 months post-discontinuation. Mucosae rarely affected (5–10%).
Management and prognosis
First-line: drug cessation (achieves resolution in 75–95%). Symptomatic relief with potent topical corticosteroids (e.g., clobetasol 0.05%) ± calcineurin inhibitors. Severe cases warrant oral prednisone (0.5–1 mg/kg tapered over 4–6 weeks) or phototherapy (NB-UVB). Recurrence risk low upon rechallenge avoidance; 26% require permanent discontinuation.
Frequently Asked Questions (FAQs)
Q: How reliable is skin biopsy in diagnosing lichenoid drug reactions?
A: Highly reliable when showing eosinophils/parakeratosis; however, 20–30% overlap with LP necessitates clinical correlation and drug history review.
Q: Can lichenoid drug reactions occur with biologics used in non-oncology settings?
A: Yes, anti-TNF agents for rheumatology/IBD frequently induce LDRs, resolving upon switching agents.
Q: What is the role of patch testing in LDR?
A: Limited utility; negative in most systemic drug reactions, positive mainly in contact lichenoid dermatitis.
Q: Do all patients need to stop the culprit drug?
A: No; 74% tolerate continuation with topical/systemic steroids, especially in oncology where benefit outweighs risk.
Q: Is direct immunofluorescence routinely needed?
A: Not routinely; reserved for DIF-positive differentials like lupus. LDR typically shows nonspecific or negative findings.
References
- Lichenoid Drug Eruption Secondary to Apalutamide Treatment — The Hospitalist. 2023. https://blogs.the-hospitalist.org/content/lichenoid-drug-eruption-secondary-apalutamide-treatment
- Cutaneous lichenoid drug eruptions: A narrative review — PubMed/NCBI. 2023-01-12. https://pubmed.ncbi.nlm.nih.gov/36652271/
- Cutaneous lichenoid drug eruptions: A narrative review evaluating… — Journal of the European Academy of Dermatology and Venereology (Wiley). 2024. https://onlinelibrary.wiley.com/doi/10.1111/jdv.18879
- Lichenoid tissue reaction/interface dermatitis: Recognition… — Indian Journal of Dermatology, Venereology and Leprology. 2018. https://ijdvl.com/lichenoid-tissue-reaction-interface-dermatitis-recognition-classification-etiology-and-clinicopathological-overtones/
- Lichenoid drug eruption — DermNet NZ. 2023. https://dermnetnz.org/topics/lichenoid-drug-eruption
- Drug-induced lichen planus — American Academy of Allergy, Asthma & Immunology (AAAAI). 2023. https://www.aaaai.org/allergist-resources/ask-the-expert/answers/old-ask-the-experts/lichen
Read full bio of Sneha Tete