Nemolizumab: Treatment for Atopic Dermatitis
Discover how nemolizumab targets IL-31 to treat moderate-to-severe eczema effectively.
Nemolizumab: Uses, Benefits and Side-effects
Introduction
Nemolizumab is a biological therapy used to treat moderate to severe prurigo nodularis (also called nodular prurigo) and atopic dermatitis. This innovative monoclonal antibody represents a significant advancement in dermatological treatment, offering targeted relief for patients with chronic inflammatory skin conditions that have proven difficult to manage with conventional therapies. The development of nemolizumab marks a paradigm shift in how clinicians approach pruritus-driven skin diseases by targeting a specific immune pathway responsible for itching and inflammation.
Nemolizumab received its first approval in Japan in 2022 (Mitchga®) for those aged 13 years and over to treat severe atopic dermatitis. This approval demonstrated the safety and efficacy profile of nemolizumab in clinical practice. In 2019, the U.S. Food and Drug Administration (FDA) awarded Galderma with a Breakthrough Therapy designation to investigate nemolizumab in treating pruritus associated with prurigo nodularis, recognizing its potential to address an unmet medical need. This designation expedites the development and regulatory review process for drugs that demonstrate substantial improvements over existing therapies.
How It Works
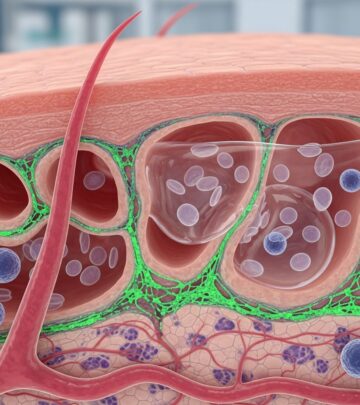
Nemolizumab is a monoclonal antibody that acts as an interleukin 31 (IL-31) antagonist. Understanding its mechanism of action requires knowledge of the immune pathways involved in atopic dermatitis. IL-31 is a cytokine produced by T-helper 2 (Th2) immune cells that plays a crucial role in the pathogenesis of atopic dermatitis. Elevated IL-31 levels are directly correlated with increased pruritus severity.
By blocking IL-31 receptor A, nemolizumab interrupts the signaling cascade that drives both itching and inflammation. Clinical trials have shown that nemolizumab effectively disrupts the itch-scratch cycle in chronic pruritic skin conditions. This mechanism is particularly important because the itch-scratch cycle perpetuates skin damage and inflammation, creating a self-sustaining problem in patients with atopic dermatitis. By blocking this pathway, nemolizumab not only reduces pruritus but also prevents the secondary inflammation caused by scratching.
The IL-31 inhibition specifically targets a pathway unique to pruritus, distinguishing nemolizumab from other biologic therapies that focus on broader immune modulation. This targeted approach means patients may experience relief from itching while maintaining immune function necessary for other protective responses.
Uses
Nemolizumab is approved and under investigation for several chronic inflammatory skin conditions:
- Atopic Dermatitis (Eczema): Moderate-to-severe cases in adolescents and adults
- Prurigo Nodolaris: Severe, therapy-resistant cases characterized by intensely pruritic nodules
- Chronic Pruritus: Other chronic pruritic conditions under clinical investigation
The primary indication is for patients with moderate-to-severe disease who have inadequately responded to topical therapies or require systemic treatment. Nemolizumab appears particularly valuable for patients with severe or therapy-resistant disease who have exhausted conventional treatment options.
Dosage and Administration
Nemolizumab is administered as a subcutaneous injection. The approved dosing regimen is as follows:
- Initial Loading Dose: 60 mg administered subcutaneously
- Maintenance Dose: 30 mg or 60 mg depending on patient weight and response
- Frequency: Administered every 4 weeks
In clinical trials evaluating atopic dermatitis, patients typically received a loading dose of 60 mg followed by 30 mg every 4 weeks for the initial treatment period. Some trials used a loading dose of 60 mg followed by either 30 mg or 60 mg maintenance doses based on baseline weight. The flexible dosing approach allows clinicians to tailor treatment to individual patient needs and response patterns.
Patients typically use nemolizumab in combination with background topical corticosteroids or topical calcineurin inhibitors, though it can also be used as monotherapy. The subcutaneous administration allows for convenient home administration, improving patient adherence and quality of life.
Benefits
Clinical trials have demonstrated significant benefits of nemolizumab treatment for patients with atopic dermatitis and prurigo nodularis:
Rapid Itch Relief
Nemolizumab reduces pruritus rapidly and improves skin changes and quality of life symptoms relative to placebo in AD patients. Improvements in pruritus have been observed as early as week 1 of treatment. Some patients experience dramatic itch relief within days of initiating therapy, with some reporting complete itch relief (0/10 on itch scales) by day 2 of treatment.
Skin Clearance
Nemolizumab decreases PVAS and EASI across a variety of doses and schedules. EASI (Eczema Area and Severity Index) and PVAS (Pruritus Visual Analog Scale) are standardized measures of disease severity and itch intensity. Clinical improvements in skin lesions and inflammation are evident by 4 weeks of treatment, with continued improvement through 12 weeks and beyond.
Quality of Life Improvements
Beyond skin clearing, nemolizumab significantly improves multiple quality of life parameters. Patients receiving nemolizumab demonstrated significant improvements in:
- Sleep disturbance and quality of sleep
- Pain and skin discomfort
- Anxiety and depression symptoms
- Overall global disease assessment
- Daily functioning and activity participation
In extended studies, more than two-thirds of patients became itch-free or nearly itch-free and 90% reported clinically meaningful improvement in quality of life.
Combination Therapy Benefits
Nemolizumab achieves a greater and more rapid improvement of PVAS and EASI in combination with topical treatments in moderate-to-severe disease than topical treatments alone. This synergistic effect makes nemolizumab an excellent option for patients inadequately responding to topical therapy alone.
Use in Specific Populations
Nemolizumab has been studied in adolescent populations with atopic dermatitis. In an open-label phase II clinical trial, 20 adolescent patients with a mean age of 14.8 years and moderate to severe atopic dermatitis received treatment with nemolizumab and demonstrated safety and efficacy in this younger population. The approval in Japan extends use to patients aged 13 years and older, suggesting potential for pediatric use in older adolescents. However, additional safety and efficacy data in younger children remains limited.
For adult patients, nemolizumab appears equally effective across age groups and has been extensively studied in phase 3 clinical trials evaluating efficacy, safety profile, and optimal dosing for moderate-to-severe atopic dermatitis.
Adverse Effects
The most common side effects of nemolizumab treatment were the common cold, worsening of eczema and an increased muscle marker (creatinine phosphokinase). More specifically, commonly reported adverse effects include:
- Nasopharyngitis (upper respiratory infection)
- Increased creatinine kinase levels
- Atopic dermatitis exacerbation
- Peripheral edema
- Injection site reactions
The adverse effects associated with nemolizumab are tolerable, making it a promising treatment of choice for moderate-to-severe AD patients. Most adverse effects are mild to moderate in severity and do not require discontinuation of therapy. Injection site reactions are typically minimal and resolve without intervention. Increased creatinine kinase levels are monitored but rarely cause clinical symptoms or require treatment cessation.
The safety profile of nemolizumab compares favorably to other biologic therapies used in atopic dermatitis, with nemolizumab appearing to be an effective treatment with an acceptable safety profile for AD.
Contraindications
While specific absolute contraindications for nemolizumab are limited, treatment should be carefully considered in patients with:
- Known hypersensitivity to nemolizumab or any excipient
- Active serious infections or immunosuppressed states
- Pregnancy and breastfeeding (safety not established)
- Patients requiring live vaccines during treatment
Patients with a history of recurrent infections or immunocompromise should be carefully evaluated before initiation of nemolizumab therapy, as IL-31 may play a role in immune response to certain pathogens.
Clinical Trial Evidence
Nemolizumab demonstrated high efficacy in the treatment of PN and AD in phase 3 trials, yielding marked improvements in symptom control with an overall favorable safety profile. In key clinical trials, 183 patients were randomly chosen to receive nemolizumab while 91 patients were given placebo. Primary endpoints included improvement in Investigator’s Global Assessment (IGA) scores and EASI-75 (75% improvement in EASI), with secondary endpoints measuring itch response and sleep quality improvements.
The clinical efficacy demonstrated in these trials supports nemolizumab’s designation as a Breakthrough Therapy. The IL-31 inhibitor, nemolizumab appears to offer effective, sustainable improvements in AD symptoms and is well-tolerated with mild AEs.
Frequently Asked Questions (FAQs)
Q: How quickly does nemolizumab work?
A: Nemolizumab works rapidly, with many patients experiencing noticeable improvement in itching within the first week of treatment. Some patients report dramatic itch relief within the first few days. Skin lesions typically show improvement by week 4, with continued improvement through 12 weeks and beyond.
Q: Can nemolizumab be used with other treatments?
A: Yes, nemolizumab is typically used in combination with topical corticosteroids or topical calcineurin inhibitors and shows superior results when combined with these treatments compared to topical therapy alone. It can also be used as monotherapy in some cases.
Q: Is nemolizumab approved by the FDA?
A: Nemolizumab received FDA Breakthrough Therapy designation in 2019 for prurigo nodularis and has been approved in Japan since 2022 for atopic dermatitis in patients aged 13 and older. Additional FDA approvals for other indications are under review as clinical trials continue.
Q: What is the cost and insurance coverage for nemolizumab?
A: As a biologic therapy, nemolizumab is typically expensive. Coverage varies by insurance plan and geographic location. Patients should consult with their healthcare provider and insurance company regarding coverage options and patient assistance programs.
Q: Are there any drug interactions with nemolizumab?
A: As a monoclonal antibody, nemolizumab has minimal drug interactions. However, live vaccines should be avoided during treatment, and patients on immunosuppressive medications should be monitored carefully. Discuss all current medications with your healthcare provider before starting nemolizumab.
Q: How long do patients need to continue nemolizumab treatment?
A: Duration of treatment varies by individual response and disease severity. Most clinical trials evaluated treatment for 16 weeks initially, with extended follow-up studies continuing for 52-56 weeks. Long-term treatment duration will be determined by your dermatologist based on your response and disease control.
References
- The role of nemolizumab in the treatment of atopic dermatitis for adults — National Center for Biotechnology Information (NCBI). 2024. https://pmc.ncbi.nlm.nih.gov/articles/PMC11485815/
- Use of Nemolizumab in the Treatment of Prurigo Nodularis and Atopic Dermatitis — Skin Therapy Letter. 2023. https://www.skintherapyletter.com/atopic-dermatitis/nemolizumab-treatment-prurigo-nodularis-atopic-dermatitis/
- Nemolizumab: Uses, Benefits and Side-effects — DermNet New Zealand. Accessed 2024. https://dermnetnz.org/topics/nemolizumab
- How NEMLUVIO® Works to Treat Eczema (Atopic Dermatitis) — Galderma Official. 2024. https://www.nemluvio.com/eczema/about-nemluvio/how-nemluvio-works
- Nemolizumab Demonstrates Marked Efficacy in Challenging Cases of Chronic Pruritus — Journal of Cutaneous and Aesthetic Dermatology Online. 2023. https://jcadonline.com/nemolizumab-chronic-pruritus/
- Nemolizumab: Uses, Interactions, Mechanism of Action — DrugBank. 2024. https://go.drugbank.com/drugs/DB15252
Read full bio of Sneha Tete