Omalizumab For Urticaria: Dosing, Efficacy, Safety
Effective biologic treatment for chronic spontaneous urticaria refractory to antihistamines, with detailed mechanisms, dosing, and safety data.
Omalizumab for Urticaria
Omalizumab, marketed as Xolair, is a recombinant humanized monoclonal anti-IgE antibody approved for treating chronic spontaneous urticaria (CSU) in patients whose symptoms persist despite H1-antihistamine therapy. It targets free IgE to interrupt mast cell and basophil activation, providing rapid symptom relief in refractory cases.
What is urticaria?
Urticaria, commonly known as hives, manifests as transient, itchy wheals (raised skin lesions) with or without angioedema (deeper swelling). Acute urticaria lasts less than 6 weeks and often resolves spontaneously, while chronic urticaria persists beyond 6 weeks. Chronic spontaneous urticaria (CSU), previously termed chronic idiopathic urticaria, affects 0.5-1% of the population, predominantly women aged 20-40, severely impacting quality of life.
CSU arises from spontaneous activation of skin mast cells and basophils, releasing histamine and other mediators. Triggers are unidentified in most cases, distinguishing it from inducible urticaria (e.g., dermatographism, cold urticaria). Autoimmune mechanisms, involving IgG antibodies against IgE or FcεRI receptors, underlie ~30-50% of CSU cases.
What is omalizumab?
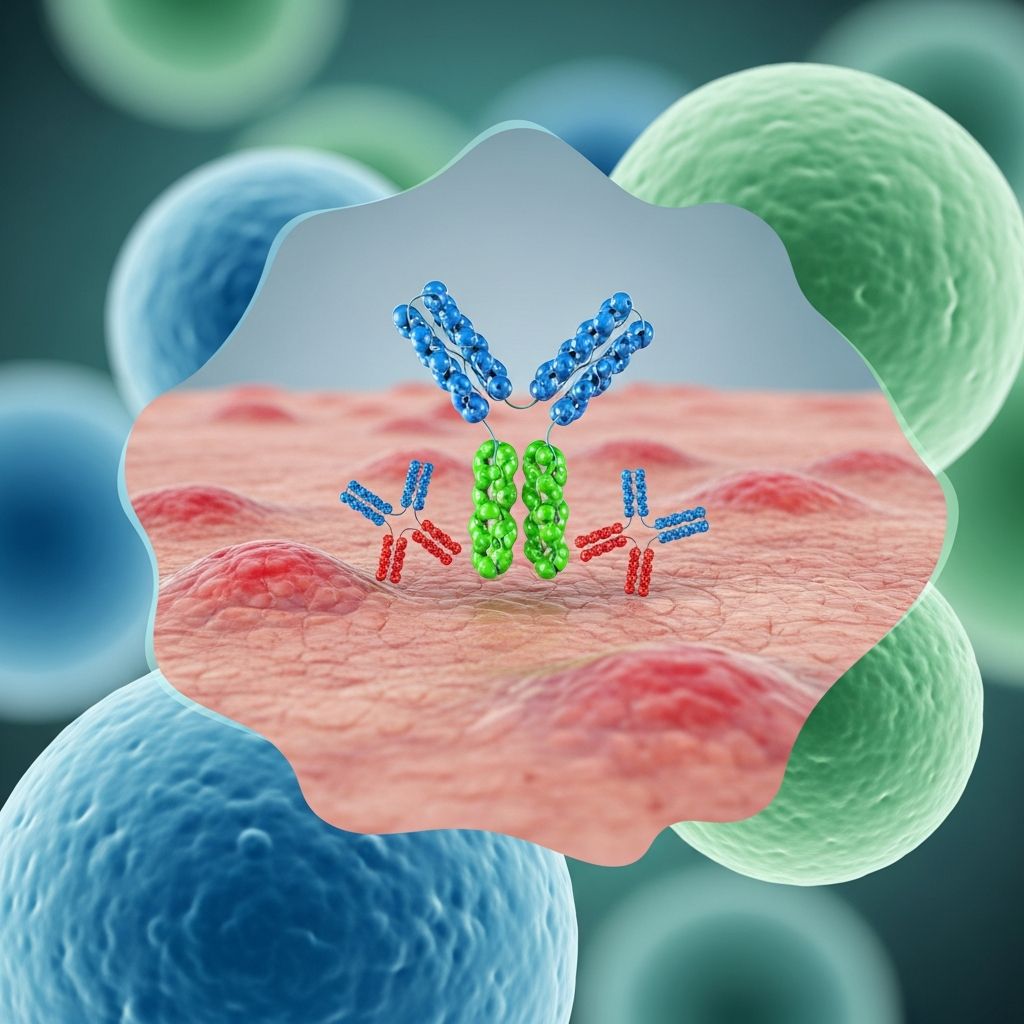
Omalizumab is a humanized IgG1 monoclonal antibody that selectively binds free circulating IgE at the Cε3 domain, preventing its interaction with high-affinity FcεRI receptors on mast cells, basophils, and dendritic cells. Administered subcutaneously every 4 weeks, it is FDA-approved for moderate-to-severe allergic asthma and CSU in adults and children ≥12 years.
Unlike polyclonal IgE, omalizumab forms inert trimers or hexamers cleared via the reticuloendothelial system without complement activation. Its pharmacokinetics show a 26-day half-life, with dosing adjusted by baseline IgE (IU/mL) and body weight (kg) for asthma, but fixed 150-300 mg for CSU.
Mechanism of action in urticaria
Omalizumab’s primary mechanism in CSU involves binding free IgE, rapidly reducing serum levels (>95% within hours) below 50 ng/mL, and downregulating FcεRI expression on effector cells by 73-99% over weeks. This blocks IgE-dependent degranulation, histamine release, and wheal formation.
Secondary effects include:
- Indirect FcεRI downregulation on basophils and mast cells due to IgE depletion.
- Reversal of basopenia and normalization of basophil histamine release in CSU patients.
- Potential modulation of IgG anti-FcεRI autoantibodies by reducing target receptor density.
- Decreased B-cell activation, TNF-α/IL-4 levels, and enhanced IFN-γ, suggesting broader immunomodulation.
While exact pathways remain incompletely understood, omalizumab addresses multifactorial CSU pathogenesis beyond simple IgE blockade.
Indications and usage
Omalizumab is indicated for CSU lasting ≥6 weeks in patients ≥12 years with symptoms inadequately controlled by H1-antihistamines (up to 4-fold dosing). It is positioned as third-line therapy per international guidelines (EAACI/GA²LEN/EDF/WAO), following failed antihistamines and before cyclosporine or immunosuppressants.
Not approved for acute urticaria or inducible urticaria subtypes, though off-label use shows variable efficacy (e.g., 50-70% response in cholinergic urticaria).
Dosing regimen
For CSU, omalizumab uses fixed dosing independent of IgE/weight:
| Dose | Frequency | Response Rate |
|---|---|---|
| 150 mg SC | Every 4 weeks | ~50-65% complete response |
| 300 mg SC (150 mg x2) | Every 4 weeks | ~75-90% complete response |
Start with 300 mg if baseline UAS7 (Urticaria Activity Score over 7 days) >28 or prior non-response to 150 mg. Median response time: 2-4 weeks; reassess at 12 weeks. Discontinue if no response by week 12. Maintenance dosing may extend to every 8 weeks in responders.
Administer by healthcare professional; pre-medicate with antihistamines if history of injection reactions.
Pharmacology
- Absorption: Peak serum levels 5-14 days post-SC; bioavailability ~62%.
- Distribution: Steady-state volume 7.3 L; no dose adjustment for mild-moderate hepatic/renal impairment.
- Metabolism: Proteolytic degradation; half-life 26 days.
- Elimination: Clearance 2.4 mL/day/kg; IgE-omalizumab complexes cleared reticuloendothelial system.
Efficacy evidence
Phase III trials (ASTERIA I/II, GLACIAL) demonstrated dose-dependent efficacy:
| Study | Dose | Complete Response (Week 12) | UAS7=0 |
|---|---|---|---|
| GLACIAL (antihistamine-refractory) | 300 mg | 74% | 52% |
| ASTERIA I | 300 mg | 53% | 36% |
| ASTERIA II | 300 mg | 66% | 44% |
Real-world data: 90% response within 3 months (n=43 retrospective). Effective in autoimmune/non-autoimmune CSU; 105+ case reports, 139 RCT patients confirm rapid, sustained remission.
Safety and side effects
Omalizumab is well-tolerated; most adverse events mild. Common (>10%):
- Injection-site reactions (45%): pain, erythema, swelling.
- Headache (15-20%).
- Upper respiratory infections (10-15%).
Rare/serious (<1%):
- Anaphylaxis: 0.1%; monitor 30-60 min post-dose.
- Urticaria exacerbation (5%).
- Hypersensitivity (anti-omalizumab IgE/IgG in 1-2%).
- Malignancy risk unconfirmed (observational data neutral).
No black-box warnings for CSU; pregnancy category B (limited data).
Monitoring
- Baseline: IgE (optional for CSU), UAS7 diary, eosinophil count.
- Follow-up: UAS7 at weeks 4,12; total IgE rises (bound complexes), not reflective of free IgE.
- Discontinue if no UAS7 improvement by week 12.
Who is it for?
Ideal candidates: Adults/adolescents with severe CSU (UAS7 >28) failing 4x antihistamines ± H2-blockers/leukotriene antagonists. Not first-line; reserve for refractory disease impacting sleep/work.
Pre-treatment work-up
- Confirm CSU diagnosis (rule out inducible/triggers).
- Baseline labs: CBC, IgE, thyroid Ab (if autoimmune suspected), autologous serum skin test.
- Assess comorbidities (asthma, nasal polyps benefit dual-indication).
- Contraindications: Hypersensitivity to omalizumab.
Special populations
- Pregnancy/Lactation: Limited data; use if benefit > risk.
- Children: Approved ≥12 years; pediatric trials ongoing.
- Elderly: No adjustment needed.
Frequently Asked Questions
What is the success rate of omalizumab in chronic urticaria?
Up to 90% achieve significant improvement; 50-75% complete remission by week 12 at 300 mg.
How quickly does omalizumab work for hives?
Median onset 1-4 weeks; some respond within days.
Can omalizumab cure urticaria permanently?
Remission persists post-discontinuation in ~60-80%; relapse possible, retreatable.
Does omalizumab require refrigeration?
Yes; store 2-8°C; allow 15-30 min room temp before injection.
Is omalizumab covered by insurance for urticaria?
Often yes with prior authorization documenting antihistamine failure.
References
- Omalizumab in the Treatment of Chronic Urticaria — Actas Dermo-Sifiliográficas. 2013-12-01. https://www.actasdermo.org/es-omalizumab-in-treatment-chronic-urticaria-articulo-S1578219013002680
- Omalizumab in Chronic Spontaneous Urticaria: Mechanisms of action — Allergy. 2016-11-19. https://onlinelibrary.wiley.com/doi/10.1111/all.13083
- Omalizumab: Allergic Asthma and Chronic Urticaria Treatment — StatPearls [Internet], NCBI Bookshelf. 2023-07-17. https://www.ncbi.nlm.nih.gov/books/NBK545183/
Read full bio of Sneha Tete