Plasmapheresis For Skin Disease: 6 Key Indications & Outcomes
Explore how plasmapheresis removes harmful plasma components to treat severe autoimmune skin conditions effectively.
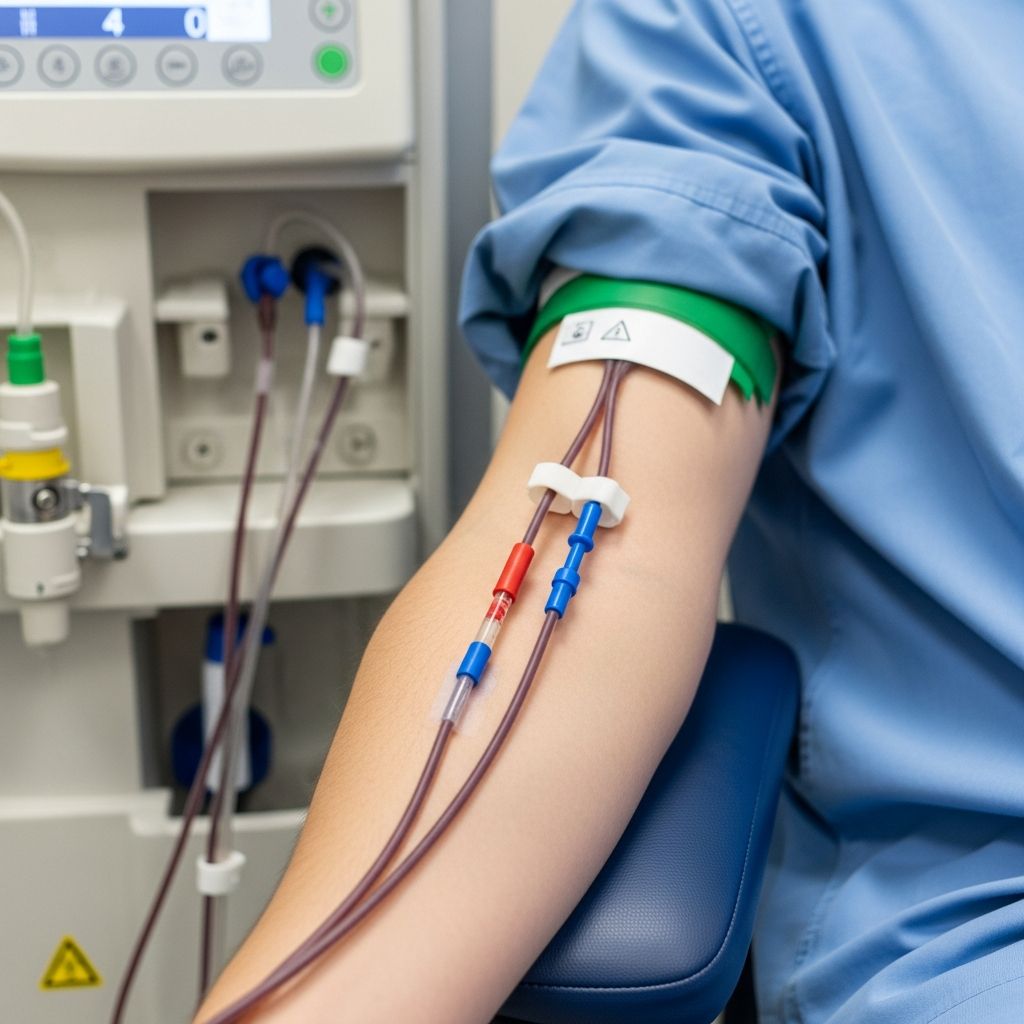
Plasmapheresis for skin disease
Authoritative facts about plasmapheresis (plasma exchange) for dermatological conditions, what it involves, who might benefit and possible side effects.
What is plasmapheresis?
Plasmapheresis, also known as therapeutic plasma exchange (TPE), is an extracorporeal blood purification technique designed to remove harmful substances from the plasma, such as autoantibodies, immune complexes, and inflammatory cytokines. The procedure involves drawing blood from the patient, separating the plasma from cellular components using a centrifuge or membrane filtration, discarding the plasma, and returning the blood cells suspended in a replacement fluid like albumin or fresh frozen plasma to the patient. This method is particularly valuable in autoimmune and inflammatory conditions where pathogenic plasma factors drive disease progression.
In dermatology, plasmapheresis targets severe skin diseases mediated by humoral immunity, providing rapid symptom relief when conventional immunosuppressants fail. Unlike dialysis, which filters small molecules, plasmapheresis selectively removes large plasma proteins responsible for tissue damage. Clinical guidelines from organizations like the American Society for Apheresis endorse its use for specific refractory dermatoses, emphasizing its role in stabilizing patients and improving outcomes.
How is plasmapheresis performed?
The plasmapheresis procedure typically occurs in a hospital or specialized apheresis unit and lasts 2–4 hours per session. Large-bore intravenous catheters are inserted into veins, often in the arms or via central lines for efficiency. Blood is anticoagulated with citrate to prevent clotting and pumped through an apheresis machine at 50–100 mL/min. Plasma is separated and removed at volumes of 1–1.5 plasma volumes per session (approximately 3–5 liters for adults), replaced to maintain oncotic pressure and volume.
Sessions are scheduled daily or every other day, with 3–10 exchanges common depending on disease severity. Monitoring includes vital signs, electrolytes, calcium levels (to counter citrate-induced hypocalcemia), and hematocrit. Post-procedure, patients rest briefly and receive supportive care like hydration and medications. Advances in automated machines have minimized risks and improved tolerability.
What skin diseases is plasmapheresis used for?
Plasmapheresis is reserved for
refractory
autoimmune blistering skin diseases where first-line therapies (corticosteroids, immunosuppressants) fail or cause intolerable side effects. Key indications include:- Pemphigus vulgaris and pemphigus foliaceus: Rapid removal of anti-desmoglein autoantibodies halts acantholysis and blister formation.
- Bullous pemphigoid: Targets anti-BP180/BP230 antibodies in elderly patients with extensive disease.
- Mucous membrane pemphigoid: For ocular or laryngeal involvement threatening vision or airway.
- Epidermolysis bullosa acquisita: Removes anti-type VII collagen antibodies.
- Toxic epidermal necrolysis (TEN)/Stevens-Johnson syndrome (SJS): Clears Fas ligand, cytokines (TNF-α, IL-6), and drug metabolites in life-threatening cases.
- Other: Dermatitis herpetiformis, linear IgA disease, paraneoplastic pemphigus.
Evidence from case series shows high response rates; for instance, in refractory TEN, plasmapheresis initiated after conventional therapy failure led to epithelial regeneration within days and full recovery after 5 sessions, with survival rates up to 91.3%. Rheumatology applications overlap, such as in lupus-related cutaneous vasculitis.
How effective is plasmapheresis?
Plasmapheresis demonstrates
high efficacy
in rapidly controlling severe skin disease activity. In pemphigus, it induces remission in 70–90% of refractory cases within 1–2 weeks, allowing steroid tapering. A review of TEN cases reported 83.3% recovery when started within 72 hours, versus longer hospital stays if delayed.| Disease | Response Rate | Avg. Sessions | Survival/Remission |
|---|---|---|---|
| Pemphigus vulgaris | 80–90% | 5–7 | High remission |
| Bullous pemphigoid | 75% | 4–6 | Rapid healing |
| Refractory TEN | 83–91% | 4.5 avg. | 91.3% survival |
| SJS/TEN spectrum | 80% | 5 | Improved LOS |
Limitations include short-term effects; relapse occurs without maintenance immunosuppression. Randomized trials are limited, but systematic reviews confirm benefits over supportive care alone. Early intervention optimizes outcomes, reducing cytokine storms and organ failure risk.
What are the risks and side effects of plasmapheresis?
Plasmapheresis is generally safe, with complication rates under 5%. Common
side effects
include:- Citrate-induced hypocalcemia (paresthesia, chills; treated with IV calcium).
- Hypotension (10–15%; managed with fluids).
- Allergic reactions to replacement fluids (rare).
- Infection at catheter sites.
- Anemia or thrombocytopenia from repeated exchanges.
Serious risks (1–2%) involve hemorrhage, embolism, or electrolyte imbalance. Contraindications include hemodynamic instability or sepsis. Long-term use risks immunoglobulin depletion, increasing infection susceptibility. Monitoring and experienced staff mitigate most issues.
What is the outcome of plasmapheresis?
Outcomes are favorable: most patients achieve disease control, shortened hospital stays (avg. 19.6 days for TEN), and steroid dose reduction. In refractory TEN, plasmapheresis reversed progression unresponsive to IVIG/TNF inhibitors, normalizing IL-6 levels and promoting re-epithelialization. Combined with immunosuppressants, it prevents rebound. Long-term prognosis depends on underlying disease management; 80–90% maintain remission with follow-up therapy.
Alternatives to plasmapheresis
Alternatives vary by disease:
- Immunoadsorption: Selective antibody removal without plasma discard; less hypotensive.
- IVIG: Blocks autoantibodies; first-line for TEN.
- Biologics: Rituximab (anti-CD20) for pemphigus; TNF inhibitors.
- High-dose steroids/immunosuppressants: Cyclophosphamide, azathioprine.
- Supportive care: Wound management for TEN.
Plasmapheresis complements these, often as rescue therapy.
Frequently asked questions about plasmapheresis for skin disease
Who is suitable for plasmapheresis?
Patients with severe, refractory autoimmune blistering diseases unresponsive to steroids and immunosuppressants. Ideal for rapid antibody clearance.
How many sessions of plasmapheresis are required?
Typically 3–10 sessions over 1–2 weeks, tailored to response. TEN often needs 4–5.
Is plasmapheresis painful?
Mild discomfort from IV access; procedure itself is painless with sedation if needed.
How much does plasmapheresis cost?
Varies; $2,000–$5,000 per session, often covered for approved indications.
Can plasmapheresis cure skin diseases?
No, it controls acute flares; maintenance therapy prevents relapse.
What preparation is needed for plasmapheresis?
Fasting not required; ensure venous access, hold anticoagulants, hydrate well.
Disclaimer: This information is for educational purposes. Consult a dermatologist for personalized advice.
References
- Plasmapheresis for refractory toxic epidermal necrolysis — Frontiers in Immunology. 2025. https://www.frontiersin.org/journals/immunology/articles/10.3389/fimmu.2025.1579349/full
- Plasma Exchange (Plasmapheresis) — American College of Rheumatology. 2023. https://rheumatology.org/patients/plasma-exchange-plasmapheresis
- Therapeutic Plasmapheresis or Plasma Exchange — Wellmark Blue Cross Blue Shield. 2023. https://digital-assets.wellmark.com/adobe/assets/urn:aaid:aem:5e0c85b3-d9fb-4211-bed2-fd44e0bc48f6/original/as/Plasma-Exchange.pdf
- Plasmapheresis/Plasma Exchange/Therapeutic Apheresis — Aetna Medical Policy. 2023. https://www.aetna.com/cpb/medical/data/200_299/0285.html
- Plasmapheresis — StatPearls, NCBI Bookshelf. 2023. https://www.ncbi.nlm.nih.gov/books/NBK560566/
- Plasmapheresis (Plasma Exchange): Therapy, Procedure — Cleveland Clinic. 2023. https://my.clevelandclinic.org/health/treatments/24197-plasmapheresis-plasma-exchange
Read full bio of medha deb