Radiation Dermatitis Pathology: 4 Essential Histologic Findings
Comprehensive pathological analysis of radiation dermatitis, detailing acute and chronic skin reactions to ionizing radiation in cancer therapy.
Radiation Dermatitis Pathology
Radiation dermatitis, also known as radiodermatitis, represents a prevalent cutaneous adverse effect of ionizing radiation exposure, particularly during radiotherapy for malignancies. This condition arises from direct damage to epidermal and dermal structures, manifesting as acute inflammatory reactions or chronic fibrotic changes. Pathologically, it involves keratinocyte apoptosis, vascular endothelial injury, and inflammatory infiltration, with severity dictated by radiation dose, fractionation, and host factors.
Introduction
Ionizing radiation induces dermatitis by generating reactive oxygen species that damage DNA in rapidly dividing cells like basal keratinocytes and endothelial cells. Acute radiation dermatitis typically emerges within 90 days of exposure, progressing from erythema to desquamation or ulceration, while chronic forms develop months to years later, featuring atrophy and telangiectasia. Nearly all patients undergoing radiotherapy experience some degree of skin reaction, with breast, head and neck, and skin cancer treatments posing highest risks.
The histopathological hallmark is basal cell vacuolization and dyskeratosis, evolving to full-thickness epidermal necrosis in severe cases. Understanding these changes is crucial for grading, prevention, and management strategies.
Clinical Features
Acute radiation dermatitis onset varies from days to weeks post-irradiation, confined sharply to the treatment field. Initial signs include faint
erythema
due to capillary dilation, followed by edema, dry desquamation, and potentially moist desquamation with exudate. Symptoms encompass pruritus, burning, and pain, peaking 2-4 weeks into therapy.- Grade 1: Faint erythema or dry desquamation.
- Grade 2: Moderate erythema or patchy moist desquamation, typically in skin folds.
- Grade 3: Confluent moist desquamation involving >1.5 cm².
- Grade 4: Skin necrosis or ulceration.
Chronic features include poikiloderma, dyspigmentation, telangiectasias, and fibrosis, appearing after cumulative doses exceeding 40-50 Gy.
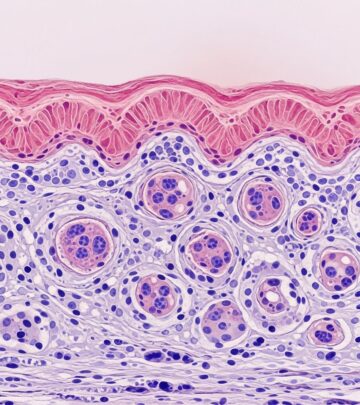
Histopathology
Histological examination reveals a spectrum of changes reflecting radiation cytotoxicity. Early lesions show subtle epidermal alterations: keratinocyte
atypia
, nuclear enlargement, and apoptosis marked by pyknotic nuclei. Basal vacuolization progresses to dyskeratotic cells sloughing into the stratum corneum.Acute Phase (1-4 weeks):
- Edema in epidermis and dermis.
- Endothelial swelling and fibrin thrombi in vessels.
- Inflammatory infiltrate of lymphocytes and histiocytes.
- TUNEL-positive apoptotic cells in keratinocytes and endothelium (up to 30/HPF in severe cases).
Subacute Phase: Full-thickness epidermal necrosis with parakeratosis, dermal edema, and vascular ectasia. CD8+ T cells predominate, mediating cytotoxicity (higher in intensity-modulated techniques like tomotherapy).
| Phase | Key Histological Features | Severity Correlation |
|---|---|---|
| Early Acute | Basal vacuolization, apoptosis | Grade 1-2 |
| Peak Acute | Epidermal necrosis, moist desquamation | Grade 3 |
| Chronic | Dermal fibrosis, atypical fibroblasts, telangiectasia | Permanent changes |
Immunohistochemistry highlights TGF-β expression in dermis (elevated in high-dose regimens), CD4/CD8 T cells, and reduced Ki-67 proliferation index.
Pathophysiology
Radiation inflicts double-strand DNA breaks, triggering p53-mediated apoptosis within 1-5 cell cycles. Basal keratinocytes, with high turnover, are most vulnerable, leading to barrier dysfunction. Vascular damage causes endothelial apoptosis, thrombosis, and ischemia, exacerbating epidermal loss.
Cytokine release (IL-1, TNF-α) amplifies inflammation; TGF-β drives fibrosis chronically. Studies show tomotherapy yields more TUNEL+ cells and CD8+ infiltrates than conventional RT, correlating with rapid grade 2 onset at 20-30 Gy skin dose.
Host factors like smoking, diabetes, and prior sun damage sensitize skin via impaired repair. Radiation recalls from chemotherapy (e.g., doxorubicin) mimic pathology through enhanced cytotoxicity.
Grading Systems
The National Cancer Institute Common Terminology Criteria (CTCAE) standardizes acute grading:
- Grade 1: Faint erythema/dry desquamation; no intervention needed.
- Grade 2: Moderate-brisk erythema/patchy moist desquamation; non-adherent dressing.
- Grade 3: Confluent moist desquamation; oral antibiotics if infected.
- Grade 4: Life-threatening necrosis; hospitalization.
Chronic grading assesses fibrosis, ulceration, and contracture. Tools like RTOG/EORTC scales aid research consistency.
Differential Diagnosis
Radiation dermatitis mimics infections (bacterial/fungal), contact dermatitis, or recall reactions. Sharply demarcated fields, temporal link to RT, and histopathology (radiation-specific atypia sans atypia in infections) distinguish it. Erythema multiforme or Sweet syndrome may overlap but lack vascular changes.
Management
Prevention emphasizes gentle skin care: aqueous creams, silicone dressings, avoid irritants. Prophylactic steroids or sucralfate reduce grade 2+ incidence.
Treatment is supportive:
- Mild: Emollients, topical steroids.
- Moderate-Severe: Barrier films, silver sulfadiazine for exudate, pain control.
- Chronic: Vascular lasers for telangiectasia, pentoxifylline for fibrosis.
Dose modification (skin sparing) in high-risk sites prevents escalation.
Prognosis
Acute reactions heal in 2-4 weeks with care; hyperpigmentation lingers. Chronic changes are irreversible but manageable. Severe grade 4 may delay therapy or necessitate breaks, impacting oncologic outcomes.
Frequently Asked Questions (FAQs)
Q: How soon does radiation dermatitis appear?
A: Symptoms start 1-2 weeks into therapy, peaking at 3-4 weeks, though erythema may delay hours-days post-exposure.
Q: Is radiation dermatitis preventable?
A: Partially, via skin-sparing techniques, prophylactic creams, and hygiene; not fully avoidable in curative doses.
Q: What does grade 3 radiation dermatitis look like?
A: Extensive moist desquamation >1.5 cm², serous exudate, high infection risk requiring dressings.
Q: Can chronic radiation dermatitis be reversed?
A: Fibrosis and atrophy persist, but symptoms improve with moisturizers, lasers, and antioxidants.
Q: Who is at highest risk for severe reactions?
A: Breast cancer patients, smokers, diabetics; head/neck sites due to folds.
Recent Advances
Modern RT like IMRT/tomotherapy reduces volume but may intensify local damage via prolonged low-dose exposure. Hydrogel dressings and growth factors (e.g., epidermal GF) show promise in trials for faster healing.
References
- Radiation Dermatitis | WoundSource — WoundSource. 2023. https://www.woundsource.com/patientcondition/radiation-dermatitis
- The Clinical Features and Pathophysiology of Acute Radiation Dermatitis — NIH/PMC. 2010-01-01. https://pmc.ncbi.nlm.nih.gov/articles/PMC2861255/
- Radiation Burn (Dermatitis): Symptoms & Treatment — Cleveland Clinic. 2023. https://my.clevelandclinic.org/health/diseases/21995-radiation-burns
- Radiation Dermatitis — DermNet NZ. 2023. https://dermnetnz.org/topics/radiation-dermatitis
- Cutaneous Radiation Injury (CRI): Information for Clinicians — CDC. 2023. https://www.cdc.gov/radiation-emergencies/hcp/clinical-guidance/cri.html
- Radiation Dermatitis | Clinical Keywords — Yale Medicine. 2023. https://www.yalemedicine.org/clinical-keywords/radiation-dermatitis
- Radiation Dermatitis | Fast Fact — Palliative Care Network of Wisconsin. 2023. https://www.mypcnow.org/fast-fact/radiation-dermatitis/
Read full bio of Sneha Tete