RSV Vaccine Side Effects: What to Expect
Comprehensive guide to RSV vaccine side effects, safety profile, and management strategies.
RSV Vaccine Side Effects: What to Expect and How to Manage Them
Respiratory syncytial virus (RSV) vaccines have become an important option for protecting older adults and pregnant women from serious RSV infections. As with any vaccine, understanding potential side effects is crucial for making informed health decisions. This comprehensive guide covers what you need to know about RSV vaccine side effects, including common reactions, rare serious events, and management strategies.
Understanding RSV Vaccine Options
Three main RSV vaccines are currently available in the United States: Abrysvo (Pfizer’s RSV vaccine), Arexvy (GSK’s RSV vaccine, adjuvanted), and mRESVIA (Moderna’s RSV vaccine). Each vaccine has been evaluated for safety in clinical trials, and all have shown effectiveness in preventing RSV disease in older adults and pregnant women. The vaccines differ slightly in their formulations and side effect profiles, which is important to understand when considering vaccination options.
Common Side Effects of RSV Vaccines
Most people who receive an RSV vaccine experience only mild side effects that resolve within a few days. These common reactions are similar to those seen with other vaccines and are generally manageable at home. Understanding what to expect can help you prepare and know when symptoms are normal.
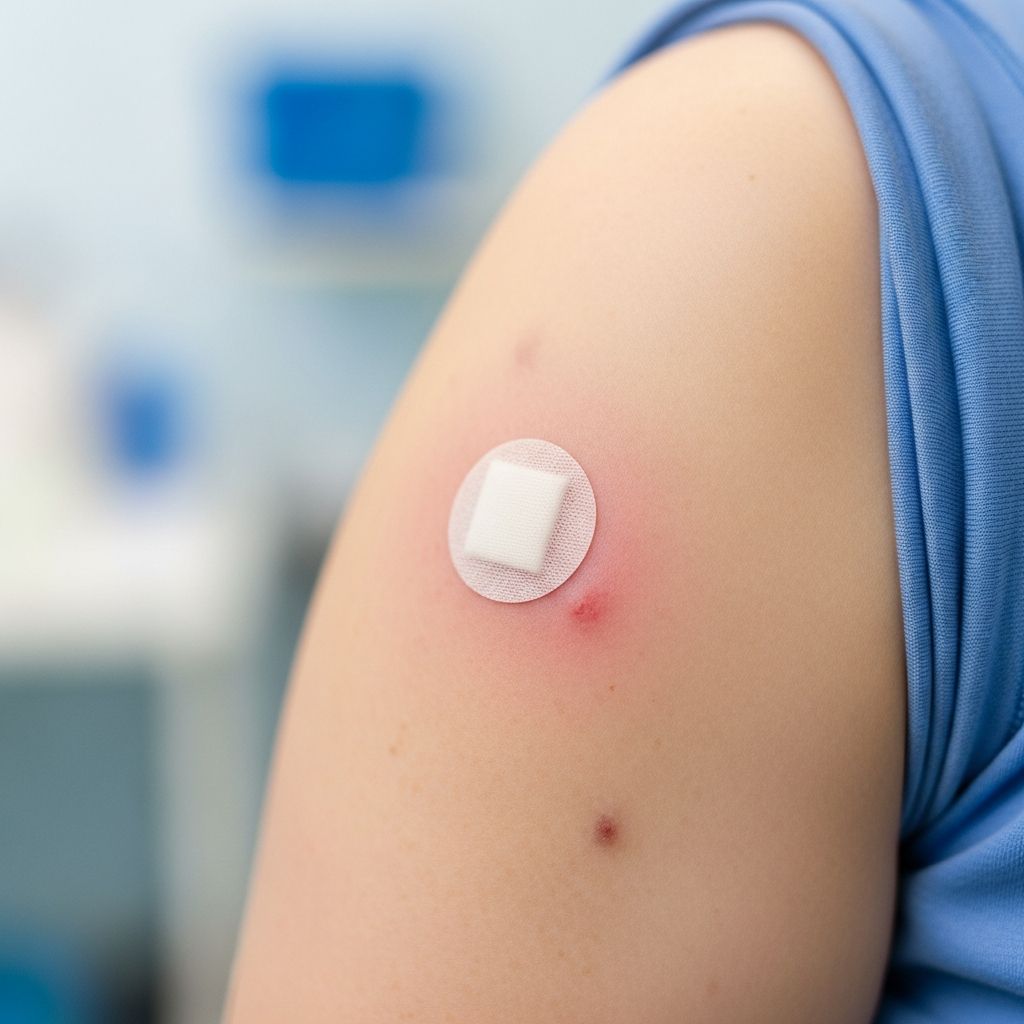
Injection Site Reactions
The most frequently reported side effects occur at the injection site. These include pain, redness, and swelling where the shot was administered. Research shows that recipients of Arexvy report injection site symptoms more frequently (29.4%) compared to Abrysvo recipients (18.9%), likely due to the adjuvant included in Arexvy that enhances immune response. These local reactions typically appear within hours of vaccination and resolve within one to two days without intervention.
Systemic Side Effects
Beyond injection site reactions, RSV vaccines can cause whole-body symptoms that reflect your immune system’s response to the vaccine. The most common systemic side effects include:
- Fatigue or tiredness – One of the most frequently reported side effects, often mild and resolving within a day or two
- Headache – Common in both vaccine recipients and clinical trial participants
- Muscle pain – Reported more frequently than joint pain during clinical trials
- Fever – Usually low-grade and temporary
- Nausea – More commonly reported in pregnant women receiving Abrysvo
- Diarrhea – Less common but still possible
- Joint pain – Mild discomfort in joints, generally not interfering with daily activities
A comprehensive analysis of 2,321 vaccine recipients found that the three most frequently reported adverse event categories were neurologic symptoms (31.6%), musculoskeletal symptoms (29.1%), and constitutional symptoms like fatigue and fever (28.4%).
Side Effects by Vaccine Type
While all three RSV vaccines show similar safety profiles overall, some differences exist between them:
| Vaccine | Key Side Effect Profile | Notable Characteristics |
|---|---|---|
| Abrysvo (Pfizer) | Pain, headache, fatigue, muscle pain | Lower injection site reaction rates; nausea more common in pregnant women |
| Arexvy (GSK) | Pain, headache, fatigue, muscle pain | Higher injection site reaction rates due to adjuvant; similar systemic side effects |
| mRESVIA (Moderna) | Injection site pain, fatigue, headache, muscle pain | No specific GBS warning; similar side effect profile to other vaccines |
Side Effects in Pregnant Women
For pregnant women receiving RSV vaccination, the most commonly reported side effects include pain at the injection site, headache, muscle pain, and nausea. Research indicates that RSV vaccines are generally well-tolerated in pregnancy, with most side effects being mild to moderate and local in nature. However, pregnant women should discuss RSV vaccination with their healthcare provider, particularly regarding timing and any individual risk factors.
Serious but Rare Side Effects
While most RSV vaccine side effects are mild and temporary, serious adverse events have been reported, though they remain rare. It is important to be aware of these potential complications:
Guillain-Barré Syndrome (GBS)
Guillain-Barré syndrome is a serious neurological condition in which the immune system attacks nerve cells, causing muscle weakness and potentially paralysis. Cases of GBS have been reported following RSV vaccination with Abrysvo and Arexvy, particularly within 42 days (6 weeks) of vaccination. Symptoms of GBS include muscle weakness, a pins-and-needles sensation, and problems with balance.
In January 2025, the FDA issued warnings about GBS based on evaluation of clinical trial data, VAERS reports, and post-marketing studies. However, the agency noted that available evidence is insufficient to confirm a causal relationship between the RSV vaccines and GBS. Among 2,321 vaccine recipients analyzed in one study, serious neuroinflammatory events including GBS were reported in 0.9% of cases.
Atrial Fibrillation
Atrial fibrillation (A-fib), an abnormal heart rhythm, was reported during clinical trials of Abrysvo and Arexvy. However, a more recent study found no increased risk of A-fib with RSV vaccines compared with other vaccines, including the flu vaccine. This suggests that the initial reports may not represent a true vaccine-related increase in risk.
Facial Paralysis
One case of facial paralysis was reported following mRESVIA vaccination. However, analysis showed no significant difference in facial paralysis cases between the vaccine and placebo groups within 28 or 42 days of vaccination, suggesting the report may not be causally related to the vaccine.
Brain and Spinal Cord Inflammation
Two rare reports of brain and spinal cord inflammation occurred in people who received Arexvy and the Fluarix Quadrivalent flu vaccine simultaneously. This represents an extremely rare event, and the connection to either vaccine individually remains unclear.
Severe Allergic Reactions
Severe allergic reactions, including anaphylaxis, are possible with any vaccine, though the risk is low. Signs of severe allergic reaction include rash, swelling in the face or throat, and difficulty breathing. Seek immediate medical attention if you experience these symptoms after RSV vaccination.
Adverse Events in Pregnancy
Some rare events have been reported among pregnant women who received RSV vaccine, including preterm birth and high blood pressure during pregnancy (pre-eclampsia). However, it remains unclear whether these events were caused by the vaccine, as these conditions can occur naturally during pregnancy. Healthcare providers should weigh the benefits of RSV protection against any individual risk factors when considering vaccination in pregnancy.
Managing RSV Vaccine Side Effects
Most RSV vaccine side effects can be effectively managed at home with simple strategies:
- Rest and hydration – Get adequate sleep and drink plenty of fluids to support your immune system and reduce fatigue
- Over-the-counter pain relievers – Acetaminophen or ibuprofen can help manage headache, muscle pain, and fever (consult your healthcare provider about appropriate dosing)
- Cold compress – Apply a cool, damp cloth to the injection site to reduce pain, swelling, and redness
- Arm movement – Gentle movement of your arm can help prevent stiffness and discomfort at the injection site
- Light meals – If experiencing nausea, eat small, light meals and avoid heavy foods
Most side effects resolve without treatment within one to two days. Contact your healthcare provider if side effects are severe, worsening, or persist beyond a few days.
When to Seek Medical Care
While most RSV vaccine side effects are minor, certain symptoms warrant immediate medical attention:
- Severe allergic reactions (difficulty breathing, swelling of face or throat, severe rash)
- Chest pain or pressure
- Severe muscle weakness or paralysis
- Difficulty walking or climbing stairs
- Tingling or numbness that spreads
- Loss of consciousness
- Any side effect that is severe or worsening
RSV Vaccine Safety Profile Overview
Overall, RSV vaccines have demonstrated a strong safety profile in clinical trials and real-world use. The adverse events observed are consistent with previous Phase II/III trial findings, with both Abrysvo and Arexvy providing safe protection against RSV for older adults, a population that previously had no vaccination option. The vaccines have been approved by the FDA and are recommended for certain populations based on age, pregnancy status, and health conditions.
Frequently Asked Questions
Q: How long do RSV vaccine side effects last?
A: Most common side effects are mild and resolve within one to two days. Injection site reactions typically appear within hours and fade quickly. Systemic side effects like fatigue and muscle pain usually improve within 24-48 hours.
Q: Are RSV vaccine side effects worse than the actual RSV infection?
A: RSV vaccine side effects are typically much milder than an actual RSV infection. In older adults and pregnant women, RSV infection can cause severe respiratory illness, hospitalization, and serious complications. Vaccine side effects are usually temporary and manageable.
Q: Which RSV vaccine has fewer side effects?
A: Abrysvo generally produces fewer injection site reactions (18.9%) compared to Arexvy (29.4%), likely due to differences in formulation. However, all three vaccines have similar overall safety profiles with mild, manageable side effects.
Q: Can I take pain relievers before getting an RSV vaccine?
A: Consult your healthcare provider before taking pre-vaccination medications. Generally, over-the-counter pain relievers can be taken after vaccination to manage side effects, but taking them beforehand may reduce vaccine effectiveness.
Q: Is it safe to get an RSV vaccine if I have a history of severe allergies?
A: Anyone with a history of severe allergy to vaccine ingredients should discuss alternatives with their healthcare provider. Most people with allergies can safely receive RSV vaccines, but individualized medical advice is important.
Q: Should pregnant women get the RSV vaccine?
A: RSV vaccines are generally well-tolerated in pregnant women with mild to moderate side effects. However, pregnant women should discuss the benefits and risks with their healthcare provider based on individual circumstances and risk factors.
Q: What is the difference between Abrysvo, Arexvy, and mRESVIA?
A: All three vaccines protect against RSV but use different technology platforms. Abrysvo uses recombinant protein technology, Arexvy uses adjuvanted recombinant protein, and mRESVIA uses messenger RNA technology. Side effect profiles are similar, though Arexvy may cause more injection site reactions.
References
- What to Know About the RSV Vaccine — Geisinger. 2023-10-24. https://www.geisinger.org/health-and-wellness/wellness-articles/2023/10/24/16/29/rsv-vaccine
- 8 RSV Vaccine Side Effects to Watch For — GoodRx. https://www.goodrx.com/conditions/rsv/rsv-vaccine-side-effects
- Adverse Events Reported Following RSV Prefusion F Protein Vaccination in Nonpregnant Adults Aged 60 Years or Older — National Center for Biotechnology Information (NCBI). https://pmc.ncbi.nlm.nih.gov/articles/PMC12731708/
- Respiratory Syncytial Virus (RSV) Vaccine Safety — Centers for Disease Control and Prevention (CDC). https://www.cdc.gov/vaccine-safety/vaccines/rsv.html
- RSV (respiratory syncytial virus) vaccine – what you need to know — MedlinePlus. https://medlineplus.gov/ency/article/007797.htm
- Side Effects Associated With Respiratory Syncytial Virus Prefusion F (RSVpreF) Maternal Vaccination: A Scoping Review — Cureus. https://www.cureus.com/articles/368597-side-effects-associated-with-respiratory-syncytial-virus-prefusion-f-rsvpref-maternal-vaccination-a-scoping-review
- RSV Vaccine Safety: Information for Healthcare Providers — Yale Medicine. https://www.yalemedicine.org/news/should-you-get-an-rsv-vaccine
Read full bio of Sneha Tete