Sickle Cell Disease: Symptoms, Causes & Treatment
Understanding sickle cell disease: Causes, symptoms, complications, and treatment options.
What Is Sickle Cell Disease?
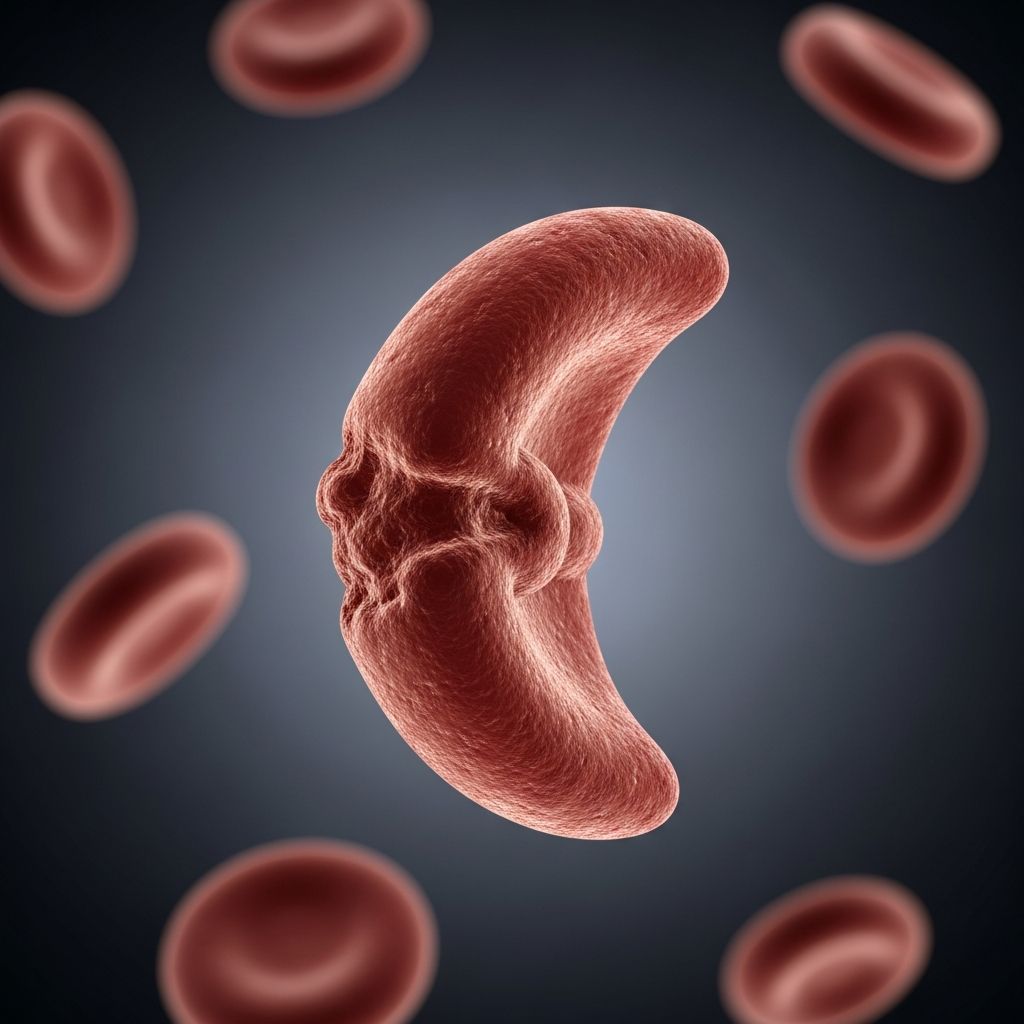
Sickle cell disease (SCD) is an inherited blood disorder caused by a change in the HBB gene that affects how your body produces red blood cells. In this genetic condition, your red blood cells form into a “C” shape—resembling a crescent moon or a farm tool called a sickle—instead of their normal round shape. These sickled cells are rigid and sticky, causing them to clump together and block blood vessels, preventing oxygen from reaching your tissues and organs. This obstruction can trigger episodes of severe pain and lead to life-threatening complications affecting multiple organ systems.
Sickle cell disease is the most common hemoglobinopathy in the United States and causes significant disease-related morbidity, including multiorgan damage, chronic anemia, and debilitating pain crises. While survival rates have improved over recent decades, individuals with SCD still have a life expectancy more than two decades shorter than the general population.
Understanding the Genetic Basis
Sickle cell disease results from a mutation at the sixth codon of the beta-globin gene, which controls hemoglobin production in red blood cells. To inherit SCD, you must receive two copies of the sickle cell gene—one from each parent. This autosomal recessive inheritance pattern means both parents must carry the gene for a child to develop the disease.
The mutation causes the production of hemoglobin S, an abnormal form of hemoglobin that polymerizes (clumps) when deoxygenated, distorting red blood cells into their characteristic sickle shape. This polymerization is the root cause of the complex, multi-organ pathophysiology associated with the disease.
Types of Sickle Cell Disease
Several genotypes of sickle cell disease exist, with varying severity levels:
–
Hemoglobin SS (Sickle Cell Anemia)
: The most prevalent and severe type, accounting for 60% to 70% of U.S. cases. Individuals with this genotype have two copies of the hemoglobin S gene and typically experience the most severe symptoms.-Hemoglobin SC
: A milder variant where individuals inherit one hemoglobin S gene and one hemoglobin C gene, resulting in less severe clinical manifestations than SS disease.-Sickle-Beta Zero Thalassemia
: Combines the sickle cell gene with a beta-thalassemia mutation. This genotype has a clinical course as severe as homozygous SCD because there is no hemoglobin A production.-Sickle-Beta Plus Thalassemia
: Another combination variant with variable severity depending on the amount of normal hemoglobin produced.While other sickle cell variants tend to have a milder clinical course, a broad range of disease severity can be observed within individual genotypes.
Symptoms and Complications
Acute Complications
Vaso-Occlusive Crises: These are the most characteristic acute complications of SCD, occurring when sickled cells block blood vessels and prevent oxygen flow. Patients experience episodes of severe pain, typically in the bones, chest, joints, and abdomen. These crises can last from hours to days and vary greatly in frequency and severity among individuals.
Acute Chest Syndrome: A life-threatening condition characterized by chest pain, fever, cough, and respiratory distress. It results from sickling in the lungs and can be triggered by infections, fat emboli, or other factors.
Stroke: Sickled cells can damage blood vessels in the brain, increasing the risk of stroke, particularly in children. Regular blood transfusions are used preventatively in high-risk patients.
Chronic Complications
Chronic Anemia: The sickled red blood cells have a shorter lifespan (10-20 days instead of the normal 120 days), leading to chronic hemolytic anemia, fatigue, and reduced oxygen-carrying capacity.
Organ Damage: Chronic sickling damages multiple organs including the kidneys, liver, heart, and lungs. Kidney disease can lead to increased urination and dehydration. Pulmonary hypertension develops from lung damage, while liver cirrhosis can result from repeated infarction.
Bone Problems: Osteonecrosis (bone death) commonly affects the femoral head, requiring surgical intervention in severe cases. Bone infarcts cause significant pain and disability.
Thrombosis: SCD is a hypercoagulable state where various mechanisms enhance platelet function, activate the coagulation cascade, and impair fibrinolysis. Venous thromboembolism affects nearly a quarter of adult patients and appears to be a risk factor for death in SCD.
Infections: Functional asplenia (reduced spleen function) increases susceptibility to bacterial infections, particularly from Streptococcus pneumoniae, Haemophilus influenzae, and Salmonella species.
Diagnosis of Sickle Cell Disease
Several diagnostic methods can identify sickle cell disease:
–
Newborn Screening
: Most U.S. states include SCD screening in newborn screening programs, allowing early detection and intervention.-Blood Tests
: Hemoglobin electrophoresis and high-performance liquid chromatography (HPLC) can identify abnormal hemoglobin patterns.-Prenatal Testing
: Healthcare providers can diagnose SCD before birth using chorionic villus sampling or amniocentesis.-Genetic Testing
: DNA testing can identify specific sickle cell genotypes and carrier status.Treatment Approaches
Supportive Care
Supportive care forms the foundation of SCD management and includes:
– Pain management with appropriate analgesics during crises- Hydration to prevent sickling- Oxygen therapy when needed- Blood transfusions for symptomatic acute anemia, stroke management, and preoperative optimization- Prophylactic penicillin and immunizations to prevent pneumococcal infections
Disease-Modifying Agents
Hydroxyurea: A disease-modifying agent that increases fetal hemoglobin production, reducing sickling and decreasing the frequency and severity of pain crises and acute complications.
L-Glutamine: Approved by the FDA in 2017, L-glutamine oral powder reduces acute complications of SCD by reducing reactive oxygen species through blockade of cellular adhesion, inhibition of hemoglobin S polymerization, and antioxidant effects.
Curative Intent Treatments
Bone Marrow Transplant: The only established potential cure for sickle cell disease, bone marrow transplantation replaces diseased bone marrow with healthy donor bone marrow (where red blood cells are made). However, finding a suitable donor match—often a close family member like a sibling—is challenging, and the procedure carries significant risks, including rejection and infection.
Gene Therapy: Gene therapies are progressing rapidly, with FDA approval of the first potentially curative gene therapies for SCD treatment. These therapies modify patient cells to produce functional hemoglobin, offering hope for long-term disease remission or cure without the need for matched donors.
Management During Crisis
When experiencing a vaso-occlusive crisis, patients can manage pain at home by taking recommended pain relievers, drinking plenty of fluids, and applying heat to the affected area. However, if pain cannot be managed at home, patients should contact their healthcare provider or go to the emergency room for professional medical intervention.
The Medical Home Model of Care
Establishing a medical home—comprehensive care based on a partnership between the patient, family, primary care physician, and other medical staff—is paramount to optimal SCD management. Typically, care is provided by a hematologist in collaboration with the primary care physician. In some cases, a single comprehensive sickle cell clinic provides coordinated care, while in other instances, a primary care physician knowledgeable in SCD care serves as the sole provider with specialist referrals as needed.
Regular medical evaluations are essential for assessing disease severity and progression. These evaluations include detailed history and physical examination to identify deviations from previous clinical status and new stressors. Annual comprehensive reviews should screen for chronic complications and facilitate specialty referrals when necessary.
Living with Sickle Cell Disease
Managing sickle cell disease requires ongoing collaboration between patients, primary care physicians, hematologists, and other healthcare specialists. Anticipatory guidance and comprehensive care planning help prevent complications and improve quality of life. Patients benefit from education about triggers that may precipitate crises, such as dehydration, infections, extreme temperatures, and stress.
Many new drugs are in development and undergoing clinical testing, offering hope for improved treatment options and potentially curative approaches in the coming years.
Frequently Asked Questions
Q: Is sickle cell disease curable?
A: Currently, bone marrow transplant is the only established cure, though it requires a matched donor and carries risks. Gene therapy represents a promising new curative approach with recent FDA approvals showing potential for long-term remission without the need for donor matching.
Q: Can you inherit sickle cell disease from just one parent?
A: No. Sickle cell disease is an autosomal recessive condition, meaning you must inherit the sickle cell gene from both parents to develop the disease. If you inherit the gene from one parent only, you become a carrier with sickle cell trait.
Q: What triggers sickle cell crises?
A: Common triggers include infection, dehydration, extreme temperature changes, stress, physical exertion, and high altitude. Identifying and avoiding personal triggers can help reduce crisis frequency.
Q: How often do sickle cell crises occur?
A: The frequency varies widely among individuals. Some people experience crises multiple times per year, while others may have few crises. Disease severity depends on the genotype and individual factors.
Q: What is the life expectancy for someone with sickle cell disease?
A: While survival has improved significantly, individuals with SCD typically have a life expectancy more than two decades shorter than the general population, though this varies based on access to care and disease complications.
Q: Are there new treatments for sickle cell disease?
A: Yes. In addition to established therapies like hydroxyurea, newer treatments include L-glutamine, which received FDA approval in 2017, and gene therapies that recently gained FDA approval, offering potentially curative options for eligible patients.
References
- Sickle Cell Disease (SCD) Symptoms, Causes & Treatment — Cleveland Clinic. 2025-08-13. https://my.clevelandclinic.org/health/diseases/12100-sickle-cell-disease
- Sickle cell disease: A primary care update — Cleveland Journal of Medicine. 2020. https://www.ccjm.org/content/87/1/19
- Targeting sickle cell disease root-cause pathophysiology with small molecules — PubMed/NIH. https://pubmed.ncbi.nlm.nih.gov/31399526/
- New Wave of Potentially Curative Treatment Offers Hope for Sickle Cell Disease — Catholic Health Association. 2025. https://www.chausa.org/news-and-publications/publications/health-progress/archives/summer-2025/new-wave-of-potentially-curative-treatment-offers-hope-for-sickle-cell-disease–how-can-we-eliminate-hurdles-to-build-on-the-promise
Read full bio of medha deb